Results for 'All Articles'
Browse oncology articles matching All Articles — expert analysis, clinical perspectives, and industry updates from across drug development and care delivery. Use the tabs above for podcasts, press releases, partners, and people for the same search.

What decision-makers actually do when they can't evaluate reasoning directly
There is a scene most advisory professionals will recognise. A recommendation that took weeks to produce. Drawing on scientific evidence, regulatory environment, market dynamics and competitor positioning gets about four minutes in a senior forum. A question or two. A decision. The people in that r…
Apr 12th • 5 mins read

The advice was good. It didn't matter.
There is a particular kind of professional frustration that doesn't have a clean name. It isn't failure — the work was sound. It isn't incompetence. The reasoning was careful, the constraints were weighed, the recommendation was defensible. It is something more specific: the experience of having…
Apr 12th • 5 mins read

Medical Affairs value isn't what you think it is
Medical Affairs functions have spent years building the case for their value. Frameworks, metrics, dashboards, impact stories, insight delivery. The work is real, the effort is genuine, and most of it misses the point entirely. Here is the problem. Value in a senior organisational setting is not wh…
Apr 1st • 5 mins read

Why leadership teams become dependent on pressure and what it costs performance
In many high-performing environments, pressure is not the exception. It becomes the operating baseline. Deadlines compress.Decisions stack.Communication tightens.Expectations increase. And over time, something subtle but important happens. Leadership teams begin to rely on pressure to function. …
Apr 13th • 5 mins read

Reinvent innovation and become an R&D front-runner by 2030
Jan 1st • 20 mins read

The Talent Inflection Point in Pharma and Biotech: A Challenge to Companies — and a Call to Candidates
For nearly two years, the pharmaceutical and biotechnology industries have operated under an unfamiliar reality: an employer-driven hiring market. From late 2023 through most of 2025, layoffs, hiring freezes, delayed clinical programs, and constrained venture capital created one of the most diffic…
Feb 4th • 7 mins read

Large pharma companies reduced headcounts by more than 22K in 2025 as $300B patent cliff looms
Large pharmaceutical companies, each with at least $20 billion in 2025 revenue, collectively reduced their workforces by more than 22,000 employees last year. Among the 17 largest pharma companies analyzed in a Fierce Pharma review of annual reports, only five logged a head count increase in 2025…
Mar 23rd • 10 mins read

Medical Judgment at the Point of Decision
When global or regional teams rely on local medical leadership, they are often doing so under less-than-ideal conditions. Timelines are compressed. Data are incomplete. Decisions must be made within local regulatory, clinical, and organisational constraints. In these situations, the value of Medical…
Feb 12th • 5 mins read

Launch Readiness Is an Organisational Capability
Launch readiness is often treated as a technical exercise. Timelines are established, deliverables are tracked, and readiness is assessed against predefined checklists. When problems arise post-launch, the explanation is often that something was missed or delayed. In practice, most launch issues ar…
Feb 12th • 5 mins read

Business Acumen Is Not Strategic Judgment
In recent years, business acumen has become a commonly cited expectation of Medical Affairs leaders. The term is often used to describe an understanding of commercial drivers, organisational structure, and how pharmaceutical companies operate. While this understanding is important, it is frequentl…
Feb 11th • 5 mins read

Driving Therapeutic Impact: A Novel Model for Strategic Medical Affairs Integration
Jul 30th • 12 mins read

Leading From the Front: 3 Leadership Lessons From Mark Cuban
Welcome to Leadership Lab, a column dedicated to biopharma executives aiming to enhance their leadership skills and advance their careers. Every other month, Michael Pietrack, the practice lead for Kaye/Bassman’s pharma and biotech recruiting team and host of “The Pharmaverse Podcast,” shares …
Dec 17th • 6 mins read

Leadership Lab: 10 Ways Executives Can Stay Visible, Valuable Between Jobs
Welcome to Leadership Lab, a column dedicated to biopharma executives aiming to enhance their leadership skills and advance their careers. Every other month, Michael Pietrack, the practice lead for Kaye/Bassman’s pharma and biotech recruiting team and host of “The Pharmaverse Podcast,&rd…
Oct 22nd • 5 mins read

Leadership Lab: Take a Cue From Athletes by Working With an Executive Coach
Think of the most accomplished athletes of your lifetime—perhaps Michael Jordan, Tiger Woods or Tom Brady. These legends were the best of the best, arguably the GOATs of their sports. They had many things in common, including the topic of today’s Leadership Lab. Each of them hired a coach or per…
Feb 19th • 6 mins read

Leadership Lab: 5 Ways Biopharma Execs Can Restore Trust, Retain Talent After Layoffs
Welcome to Leadership Lab, a column dedicated to biopharma executives aiming to enhance their leadership skills and advance their careers. Every other month, Michael Pietrack, the practice lead for Kaye/Bassman’s pharma and biotech recruiting team and host of “The Pharmaverse Podcast,&rd…
Jun 25th • 4 mins read

Leadership Lab: 5 Steps for Building Your Personal Brand
Welcome to Leadership Lab, a column dedicated to biopharma executives aiming to enhance their leadership skills and advance their careers. Every other month, Michael Pietrack, the practice lead for Kaye/Bassman’s pharma and biotech recruiting team and host of “The Pharmaverse Podcast,&rd…
Aug 19th • 5 mins read

Leadership Lab: How To Spot When Employees Are About To Walk Away
Welcome to Leadership Lab, a column dedicated to biopharma executives aiming to enhance their leadership skills and advance their careers. Every other month, Michael Pietrack, the practice lead for Kaye/Bassman’s pharma and biotech recruiting team and host of “The Pharmaverse Podcast,” shares …
Oct 22nd • 7 mins read

Leadership Lab: 4 Ways Biopharma Leaders Can Prepare for Media Interviews
Welcome to Leadership Lab, a column dedicated to biopharma executives aiming to enhance their leadership skills and advance their careers. Every other month, Michael Pietrack, the practice lead for Kaye/Bassman’s pharma and biotech recruiting team and host of “The Pharmaverse Podcast,” shares …
Aug 20th • 6 mins read

Managing Emotions After Being Downsized
The biggest challenge a person faces after being downsized is the wide range of emotions that follow. Over the past 15 years, I’ve observed many individuals deal with this unfortunate circumstance, and I wanted to share what I’ve seen others do to cope. I genuinely hope this helps you or someon…
Oct 21st • 5 mins read

Practical Advice for When You're Downsized. What Activities will Pay-off after a Lay-off?
Have you or someone you know recently been downsized, laid off, or RIF’d? If so, you’re going to want to watch and share this video. When one is downsized, there is a range of emotions. Some feel betrayed and carry hurt feelings, while others fight with the fear of uncertainty and the reality…
Sep 15th • 5 mins read

Resume Advice: Tattoos and Breadcrumbs
Okay, Pietrack has finally lost it! What do tattoos and breadcrumbs have to do with resume advice? If you did a web search seeking resume advice, you would get pages of information about resume construction to format to length. So much of how a resume should be constructed is subjective or based o…
Jul 14th • 5 mins read

How to Elevate Your Career: Go from Individual Contributor to People Leader
Have you ever wondered how to elevate from an individual contributor to a leadership position? Well, it's all about picking the right elevator. Imagine your current position as an elevator, and it has stopped at the seventh floor. Despite pushing all the buttons, that elevator just will not go …
Jul 24th • 5 mins read

What Do Tennis and Job Interviewing Have in Common?
What do tennis and Job interviewing have in common? In a game of tennis, one person sends the ball over the net and the other person, sends it back, right? Well, when it comes to job interviewing, correspondence with the hiring manager is a lot like tennis. How? When you send the ball over the ne…
Apr 14th • 5 mins read

Hiring Managers: What Should You Do During a Hiring Freeze?
As we begin 2023, many companies are experiencing hiring freezes. Some hiring managers have a sense of how long the freeze will be, some don’t, but all know that it’s temporary. So, what can hiring managers do during the freeze to stay competitive in the war for talent? Hiring is a competition…
Jan 26th • 5 mins read

How to Know When to Make a Job Change
In career conversations, one of the most common questions I hear is: “How do I know if it’s time to move on from my company?” The real decision often comes down to a mix of factors that influence job satisfaction and long-term career growth. That’s why I use the CLAMPS framework. CLAMPS …
Aug 26th • 5 mins read

The Great Resignation is Now The Great Hesitation
Over the last few years, the employment market has moved through dramatic swings. In 2021 and 2022, millions of professionals left their roles in what became known as The Great Resignation. Jobs were abundant, companies were aggressively hiring, and the perceived level of risk was low. If a new role…
Nov 19th • 5 mins read

The Four Employment Agreement Questions Every Pharma Executive Must Ask
Welcome to Leadership Lab, a column dedicated to biotech executives aiming to enhance their leadership skills and advance their careers. Every other month, Michael Pietrack, the practice lead for Kaye/Bassman’s life sciences recruiting team and host of The Pharmaverse Podcast, shares a valuabl…
Dec 18th • 6 mins read

New Book Unites Oncology’s Brightest Minds To Innovate Cancer Cures
NEW YORK (September 9, 2025)—Voices of Oncology: Fostering a Collaborative Community of Experts to Accelerate Cancer Cures by Kirk V. Shepard, M.D., and Ramin Farhood, PharmD, M.B.A., brings together the insights of 33 distinguished contributors from across the oncology landscape. The boo…
Sep 9th • 5 mins read

Where Did My Interview Go Wrong?
During the course of a lengthy and involved interview process, job seekers are often left wondering where they went wrong, or why things didn’t pan out positively in their favor – with a great job offer at the end and an exciting next chapter to look forward to in their professional care…
Apr 24th • 1 min read

Mastering the First Impression
Why is a first impression so important? Mainly because it could also be your last, and only, impression on someone. Oftentimes, job seekers can fail at conveying a positive first impression, and they will unfortunately never get another chance to show who they truly are. This is an area that I feel …
Jan 29th • 2 mins read

Interview Differentiator-Be a Story Teller
When you search Google for the words “Interview Preparation,” you will come up with approximately 756,000,000 results. While I am sure you can find an abundance of valuable information, it is very difficult to navigate through all of the content available on the Internet to determine wha…
Mar 22nd • 1 min read

12 Interview Techniques to Help Land Your Next Job
Your next career move could very well be one interview away. Be sure to take every measure to prepare yourself and make the most of the opportunity. According to a recent Simply Hired survey of over 850 Managers, an overwhelming 89% said that “showing lack of preparation” will affect the…
May 12th • 1 min read

“Extreme Interviewing” – MSL Interview Tips and Insights from Medical Affairs Leaders
What is “Extreme Interviewing”? Is there such a thing? Does this term even exist in the dictionary? The short, and maybe obvious, answer is “No.” So perhaps I am guilty of coining a new term for the purpose of this article, but it is a concept that is very relevant and worth…
Jul 9th • 1 min read

Job Search Checklist for Aspiring Medical Science Liaisons
Tips to Help Break into Your First MSL Role The Medical Science Liaison (MSL) role, has become one of the most sought-after career paths in the Pharmaceutical and Biotech industry. What does it take to land a role as a Medical Science Liaison? As an MSL Recruiter, this is a question that I am…
May 17th • 2 mins read

MSL Retention – What Matters Most
As the Medical Science Liaison job market continues to become more and more competitive, attrition rates continue to increase as a result. One of the most common questions I receive from MSL Directors and Medical Affairs leaders is, “What can we do to keep our MSLs loyal and motivated?” …
Mar 15th • 2 mins read

MSL Hiring and Recruitment: 5 Ways to Support Diversity and Inclusion
While the COVID-19 pandemic wreaked havoc on many industries, Medical Science Liaison hiring was steady throughout 2020 and continues to remain strong in 2021. The one change in hiring that has become evident is the increase in recruitment practices that support diversity and inclusion. While we may…
May 19th • 2 mins read

How to Prepare for a Virtual or Video Interview
As we are quickly becoming thrust into the age of “virtual” meetings and video interviews, job seekers and industry professionals need to be completely prepared to master this new paradigm of interpersonal communication. Here are some important tips for acing your next video interview: …
Mar 19th • 1 min read

10 Ways to Make an Impact on a Phone Interview
The phone interview, sometimes referred to by Hiring Managers and Talent Acquisition teams as the “phone screen,” is most often the first step in the interview process. While most job seekers are familiar with this concept and first step, some think of it simply as a formality or a way t…
Apr 9th • 1 min read

Resume Writing and Editing Tips for Pharma Professionals
As a Professional Pharmaceutical Recruiter, I read and evaluate resumes (as well as CVs), every day. While I am not a professional resume writer, I have a very strong understanding of what a good resume or CV looks like and what should be included. More importantly, I keep track of what is most effe…
Aug 8th • 1 min read

6 Tips to Ace Your Next MSL Interview!
As a Medical Affairs-focused recruitment firm, we see MSL interview "wins" and slip-ups on a daily basis. From our experience working with candidates across all therapeutic areas and across the entire United States, I've noticed a few trends that might help you get that next interview "win" under yo…
Sep 28th • 1 min read

Presentation Nails and Fails: 7 Tips to Ace Your Next MSL Presentation
The presentation portion of the MSL interview is truly “make or break,” and more often than not, the most highly weighted category of the entire interview process. Countless Hiring Managers have passed on MSL candidates that do not perform well on their scientific presentation – re…
Oct 12th • 1 min read

Combatting the “Summer Slow Down" – MSL Job Search Tips for Slower Months
There is definitely a cyclical nature to hiring Medical Science Liaisons and Medical Affairs professionals. Typically, peak season for recruitment and hiring is from March to June, which is the time-frame in and around which MSLs collect their annual bonuses. However, Medical Science Liaison hiring …
Jun 23rd • 2 mins read

7 High-Cost Interview Mistakes that are Easy to Avoid
Is your job search taking much longer than expected? Have you gone on interviews and feel it went well, but you still didn’t get the job? Interviewing in a competitive job market can be more challenging than one might think. COVID-19 has not made the interview process any easier – instea…
Jul 28th • 1 min read

INTENTION: The Key to Achieving Your Goals
New Years resolutions and goals always seem to follow the same path with the same result. Start…no finish. Beginning…no end. I feel like way too many people make resolutions only to break them a few weeks later. I want to share a strategy that WORKS. Not only will it help you stick to …
Jan 21st • 1 min read

MSL Evolution: New Trends and Titles That May Emerge
The COVID-19 pandemic has clearly had a profound impact on Field Medical Affairs. Has the Medical Science Liaison role changed forever? What new titles, trends and opportunities might emerge as a result of the new age for Field Medical and the evolution of the Medical Science Liaison? Is virtual en…
Apr 5th • 1 min read

MSL People Skills: Top 10 Tips for Better Engagement
The cornerstone of the Medical Science Liaison role is RELATIONSHIP BUILDING. In a nutshell, better engagement equates to better relationships. The purpose of this article is to address the importance of proper business etiquette and the impact of social awareness and emotional intelligence as it re…
Apr 20th • 8 mins read

Promotability: How to Land Your Next Promotion
“Promotability” may be defined as one’s suitability for being promoted. Is it your goal to obtain a promotion this year? If so, how suitable or promotable are you for that next step? Job-seekers and professionals often ask me what steps are necessary for advancement. I have broken …
May 5th • 10 mins read

Positioning Yourself to Become an MSL in 2022
After attending the MSL Society’s 9th annual conference in Vegas Last week, I have taken some time to reflect on inquiries our team received about how to effectively step into the MSL role from a clinical, academic, or research background. Industry is a tough nut to crack, but it is absolutely…
Dec 21st • 10 mins read

What to do When a Job Offer is Rescinded
First off, there are a couple of reasons why a company might rescind a job offer. Number one, if the person gets an offer and over negotiates or handles the negotiation improperly, it may rub people the wrong way and could lead to the company rescinding the offer. More commonly, the reason why job o…
Jul 20th • 5 mins read

Virtual Oncology MSL Team Increases KOL Engagement by 120%
Case Study (US) The Challenge Client needed comprehensive, scalable and innovative model. Goal: Cost-effectively supplement field based MSL team to provide national coverage. Objective: Share newly published guidelines with KOLs and gain key strategic insights. The Approach Highly …
Sep 3rd • 1 min read

Clinical Educator Oncology Program Improves Adherence by 29%
CASE STUDY (US) The Challenge Client’s specialty product had a complicated oral therapy regimen requiring supplemental support to patients. Therapy administered for chronic life-threatening condition in which patients are generally older and polypharmic. Our Soluti…
Sep 3rd • 1 min read

Transforming oncology: Five frontiers driving progress in cancer care
From biomarker-driven breakthroughs to AI-powered early detection and a renewed commitment to equity and patient centricity, the past 12 months have seen major strides across cancer research, treatment, and communication. At Inizio, we’ve had a front-row seat to this transformation, supporting…
May 16th • 5 mins read

Expert perspectives live from ASCO® 2025
At the 2025 ASCO® Annual Meeting, three of our experts sat down with OncoDaily to share their unique perspectives in oncology – from amplifying the patient voice to unlocking real-world impact through data and AI. Watch the videos below. Matthew Hoelzle | Medical strategy, AI and real-wor…
Jul 2nd • 1 min read

Evaluation of Trials Comparing Single-Enantiomer Drugs to Their Racemic Precursors: A Systematic Review
In the US, a system of patents and market exclusivity provides manufacturers of new drugs protection against competition from generic drugs. Although this system balances higher prices from delayed generic competition with the need to promote new drug innovation, certain strategies, such as &ld…
May 6th • 18 mins read

Revamping the ever-changing landscape of drug development processes in the midst of COVID-19 pandemic
As this adage goes, cumulative scientific breakthroughs bolster progress. The beauty of standing on the shoulders of giants is to see far, capitalize on prevailing opportunities and leapfrog to greater heights. An example is the Nobel Prize in Chemistry recognizing Charpentier and Doudna’s dis…
Apr 29th • 2 mins read

Cancer, Clinical Trials, and Canada: Our Contribution to Worldwide Randomized Controlled Trials
Randomized controlled trials (RCTs) are the gold standard for evaluating the efficacy of new cancer therapies. Since widespread adoption of the RCT in the 1970s, cancer clinical trials have evolved considerably. Work by our group and others have shown that RCTs have evolved over time to become …
Apr 13th • 10 mins read

Biases in study design, implementation, and data analysis that distort the appraisal of clinical benefit and ESMO-Magnitude of Clinical Benefit Scale (ESMO-MCBS) scoring
The European Society for Medical Oncology-Magnitude of Clinical Benefit Scale (ESMO-MCBS) was first published in 2015 and revised in 2017. With a growing recognition that many cancer medicines provided modest benefits disproportionate to their high costs, the oncology community needed a tool that co…
Apr 20th • 8 mins read

Updated estimates of eligibility for and response to genome-targeted oncology drugs among US cancer patients, 2006-2020
Precision oncology relies upon genomic sequencing of a patient’s tumor to determine optimal treatment. Precision therapies typically target genetic aberrations within cancer, and this approach has widespread enthusiasm driven by high response rates. Often, genomically-targeted drugs gain Food …
Apr 20th • 7 mins read
Comparative study on anticancer drug access times between FDA, EMA and the French temporary authorisation for use program over 13 years
The cancer incidence continues to rise worldwide. There were about 18,000,000 new cases in 2018 and about 9,500,000 deaths. Despite therapeutic advances, cancer has become the leading cause of death since 2004 in France, with 157,000 deaths in 2018. To cope with this challenge, societies need to ada…
Apr 7th • 12 mins read

Virtual Clinical Trials in Oncology-Overview, Challenges, Policy Considerations, and Future Directions
COVID-19 has proven to be a transformational event for medicine in the 21st century, driving rapid multi-sectoral change (eg, care provision via telemedicine and site-of-care optimization) and creating new value propositions for healthcare systems worldwide. The potential for such change is especial…
Apr 8th • 4 mins read
Advances in basic research in oncology in 2020: Bridging basic science and clinical care
Cancer has now become a disease that can be understood and interpreted even at the genetic level, and the advent of massive parallel sequencing has ushered in an era of systematic documentation of these genomic changes encompassing the whole genome. The year 2020 had a dramatic start in this area wi…
Mar 24th • 1 min read

Are Quality of Randomized Clinical Trials and ESMO-Magnitude of Clinical Benefit Scale Two Sides of the Same Coin, to Grade Recommendations for Drug Approval?
The Grading of Recommendations Assessment, Development and Evaluation (GRADE) was published in 2001 by non-oncologic societies and later endorsed by the US Food and Drug Administration. A simplified GRADE adaptation scale offered two grades of recommendation: strong and weak. GRADE has been used by …
Feb 11th • 3 mins read

Does biomarker use in oncology improve clinical trial failure risk? A large-scale analysis
Cancer continues to be a major challenge in medicine, as it remains the second leading cause of death in the United States, after heart disease, with a forecasted 1.7 million new cases in 2018. Finding new treatments remains a challenge, as illustrated by drug failures rates during clinica…
Feb 23rd • 8 mins read

Assessment of Coverage in England of Cancer Drugs Qualifying for US Food and Drug Administration Accelerated Approval
The US Food and Drug Administration’s (FDA) accelerated approval process allows drugs to be approved based on clinical trial findings that would otherwise not be acceptable for use in the traditional FDA approval process (ie, indicating changes based on surrogate measures that are only reasona…
Feb 22nd • 10 mins read

The First 2 Years of Biosimilar Epoetin for Cancer and Chemotherapy-Induced Anemia in the U.S.: A Review from the Southern Network on Adverse Reactions
Biosimilar drugs, close copies of patented biologicals, are intended to provide access to less expensive, highly similar versions of approved reference biological agents. The biological epoetin accounts for $1.8 billion in drug spending annually worldwide, primarily for treatment of anemia due …
Mar 12th • 7 mins read

Assessment of Food and Drug Administration- and European Medicines Agency-Approved Systemic Oncology Therapies and Clinically Meaningful Improvements in Quality of Life: A Systematic Review
For patients with cancer treated with palliative intent, quality of life (QOL) is a critical aspect of treatment decision-making, alongside survival. In some cases, patients with advanced cancer have even prioritized QOL over survival. However, submission of QOL data are not mandatory for oncol…
Feb 11th • 4 mins read

Accelerated drug approvals in oncology: Pros and cons
Any drug or pharmaceutical product is approved for marketing once it succeeds in the adequate and well-controlled phase III trial. Marketing approval of drugs, provided by the US Food and Drug Administration (FDA), could be granted provided the safety, as well as efficacy measures, has been taken ca…
Sep 14th • 4 mins read
Finding the Right Drug at the Right Dose the First Time: Has the Era of Personalized Formularies Finally Arrived?
Every day, millions of Americans take medications that will never work for them fully as intended. Some patients will see little to no benefit. Others may have a variety of adverse reactions, including potentially fatal reactions. This is because individuals metabolize drugs differently, and, in man…
Sep 27th • 1 min read
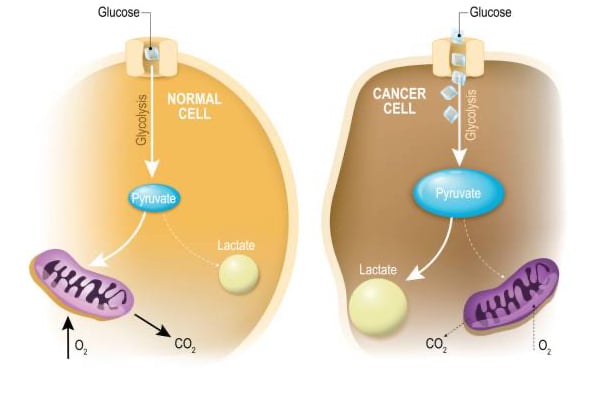
“Oncometabolism: The switchboard of cancer: An editorial”
In the 1920’s, Otto Warburg, one of the greatest biochemists of all time, uncovered an intriguing bioenergetic phenotype shared by most tumor cells: a higher than normal reliance on lactic acid fermentation for energy generation. Warburg’s seminal finding gave rise to a new field of canc…
Feb 1st • 1 min read
Loose Regulatory Standards Portend a New Era of Imprecision Oncology
Over the last 20 years, precision oncology has changed the treatment landscape for patients with specific cancers and has become a much sought-after goal in oncology drug development. However, recent drug approvals by the United States Food and Drug Administration (FDA) under the auspices of precisi…
Dec 1st • 4 mins read
Confounding factors in exposure–response analyses and mitigation strategies for monoclonal antibodies in oncology
While contemporary drug development in oncology strives to deliver novel therapies to patients rapidly, it is also important to optimize dosing regimens to improve patient-centered care. Doses selected for pivotal trials may be efficacious doses, but not necessarily optimal to minimizing toxicity an…
Nov 20th • 12 mins read
A narrative review of biosimilars: a continued journey from the scientific evidence to practice implementation
The simplest forms of biologic drugs entered the U.S. market in the 1970s and primarily consisted of blood products and vaccines. The rise of cloning and gene expression technology enabled biosynthesis of genetically modified organisms, which allowed the production of increasingly complex biologic m…
Aug 3rd • 10 mins read

First person profile: William Sellers, MD
Based in Cambridge, Massachusetts, Dr. Sellers is a core member of the Broad Institute of MIT and Harvard, a senior advisor to the president for experimental therapeutics at Dana-Farber Cancer Institute, and a professor of medicine at the Dana-Farber Cancer Institute and Harvard Medical School. He i…
Nov 6th • 2 mins read

Safeguarding cancer research funding by European charities amidst the COVID-19 pandemic
The COVID‐19 pandemic is having severe consequences for cancer patient care. Cancer patient screening and referrals are being delayed, ongoing clinical trials are being brought to a halt, and new ones are not being approved. Guidelines and clinical recommendations for cancer patients have already …
Nov 22nd • 3 mins read

Seven decades of chemotherapy clinical trials: a pan-cancer social network analysis
The modern era of chemotherapy began in 1946, with publications describing therapeutic uses of nitrogen mustard. Over the next 70 years, the repertoire of available cancer treatments has expanded at an ever-increasing pace. Chemotherapeutics have a notably low therapeutic index, i.e., the diffe…
Oct 16th • 12 mins read

Advances and Challenges in Pediatric and Childhood Cancers
While risk factors for gene mutations responsible for several adult cancers have been identified, they remain largely unknown in childhood malignancies. Fortunately, relative to adult cancers, pediatric cancers are uncommon. Leukemia, central nervous system (CNS) tumors, lymphoma, Wilms' tumor, and …
Jun 27th • 2 mins read

Liquid biopsy in oncology: a consensus statement of the Spanish Society of Pathology and the Spanish Society of Medical Oncology
By the year 2030, 22.2 million new cases of cancer are expected worldwide: a challenge for cancer patient diagnosis and therapeutic approaches. Despite this increase, patient prognosis has improved with a gradual decrease in cancer-related mortality, reflecting the breakthroughs in early diagnosis a…
Sep 26th • 17 mins read

Leveraging existing data to contextualize phase II clinical trial findings in oncology
More than 250 000 women are diagnosed with early-stage breast cancer (EBC) in the USA each year. Of these, up to 30% have amplification of the human epidermal growth factor 2 (HER2).2 The current standard of care for HER- positive (HER2þ) EBC is chemotherapy plus HER2-directed therapy to compl…
Sep 21st • 3 mins read
A Field Test of Major Value Frameworks in Chemotherapy of Nasopharyngeal Carcinoma-To Know, Then to Measure
The goal of any cancer therapy is to help patients live longer, or live better, or both. In the clinic, oncologists, and patients need to discuss the balance of benefit and toxicity associated with different treatment options, to make the best decision for each patient. The European Society for Medi…
Aug 12th • 10 mins read

Application of the ESMO-Magnitude of Clinical Benefit Scale (V.1.1) to the field of early breast cancer therapies
As the population ages, the incidence and prevalence of cancer are expected to continue to rise both in developed and developing countries. The estimated total annual economic cost of cancer was US$1.16 trillion in 2010, about 2% of global gross domestic product and is continuing to rise exponential…
Sep 6th • 20 mins read
Lessons learnt from scoring adjuvant colon cancer trials and meta-analyses using the ESMO-Magnitude of Clinical Benefit Scale V.1.1
Colorectal cancer is the third most common tumour in men, the second in women and second place in cancer-related cause of death in the world. Mortality has declined over the years for several reasons, including colorectal cancer screening and more effective systemic therapies in both the adjuvant se…
Sep 6th • 17 mins read

Professional Medical Writer Assistance in Oncology Clinical Trials
Academic publishing remains the cornerstone of cancer research and scientific communication of clinical trials. Over the years, there has been increasing interest in the use of professional medical writers (PMWs) to help investigators improve quality of writing and/or reduce the time to publication.…
Sep 17th • 7 mins read
Quantitative Clinical Pharmacology of T‐Cell Engaging Bispecifics: Current Perspectives and Opportunities
Advancements in antibody engineering and recent clinical successes have led to enthusiasm for the development of bispecific modalities with the unique ability to bind to two distinct antigens or two different epitopes on the same antigen. T‐cell directing/engaging bispecific agents (TDBs), in…
Nov 18th • 15 mins read
Clinical benefit of immune checkpoint inhibitors approved by US Food and Drug Administration
Knowledge of the potential benefits and risks associated with the use of anticancer therapies is fundamental for making treatment-related recommendations and decisions. Two important oncology societies have recently taken a step forward to quantize the clinical benefit. The American Society of Clini…
Aug 31st • 16 mins read

Past, Current, and Future Cancer Clinical Research Collaborations: The Case of the European Organisation for Research and Treatment of Cancer
Academia-industry collaborations in the discovery stage of the pharmaceutical research and development process have led to major scientific advances that have benefited patients significantly. Their rationale and challenges are well-documented, and models for such partnerships have been extensively …
Aug 16th • 8 mins read

Pediatric Oncology Clinical Trials and Collaborative Research in Africa: Current Landscape and Future Perspectives
By mid-2019, the population of African children age < 15 years was > 535.1 million, 41% of the total population. Although infectious diseases, malnutrition, and neonatal deaths are the main causes of childhood mortality on the continent, noncommunicable diseases such as childhood cancer …
Aug 7th • 10 mins read

Clinical benefit and cost of breakthrough cancer drugs approved by the US Food and Drug Administration
Improved understanding of the molecular basis of cancer has led to the discovery of several new therapies, which, in some cases, have demonstrated substantial anti-tumor activity in early phase trials and subsequently improved overall survival (OS). In 2012, the breakthrough therapy designation was …
Jul 22nd • 12 mins read
Comparison of Access to Novel Drugs for Lymphoma and Chronic Lymphocytic Leukemia Between India and the United States
Blood cancers are a significant public health problem worldwide and a leading cause of death in the United States and India. According to GLOBOCAN 2018, the annual incidence of lymphoma is 82,548 in the United States and 37,225 in India. The annual incidence of chronic lymphocytic leukemia (CLL) was…
Jul 21st • 12 mins read
Quantitative Translation in Immuno-Oncology Research and Development
Differentiating features of IO drug development include increased diversity of modalities, greater emphasis on combinations,including optimal sequencing, and patient selection strategies that require multi-dimensional characterization of the tumor micro environment. Additionally, the complexity of h…
Jul 9th • 3 mins read

Biosimilars in oncology: key role of nurses in patient education
When patients are introduced to biosimilars, as with any new therapy, they are likely to have questions as to what they are and why they are important. In this context, it may be useful to discuss how biologics are made and compare biosimilars with generic small-molecule medicines. Although bio…
Jun 15th • 10 mins read
Tumor Growth Dynamic Modeling in Oncology Drug Development and Regulatory Approval: Past, Present, and Future Opportunities
Model-informed drug development (MIDD) approaches have advanced rapidly in drug development in recent years. Additionally, the Prescription Drug User Fee Act (PDUFA) VI has specific commitments to further enhance MIDD. Tumor growth dynamic (TGD) modeling, as one of the commonly utilized MIDD approac…
Jun 26th • 18 mins read

Clinical development success rates and social value of pediatric Phase 1 trials in oncology
Phase 1 clinical trials in oncology are designed to test safety, identify dose recommended for further testing and probe the pharmacologic and pharmacodynamic performance of new treatments. The probability that new cancer drugs tested in Phase 1 will reach regulatory approval is reported to be aroun…
Jun 21st • 28 mins read
Characterizing Exposure–Response Relationship for Therapeutic Monoclonal Antibodies in Immuno-Oncology and Beyond: Challenges, Perspectives, and Prospects
Analysis of the exposure response (E–R) relationship has long been an important tool for dose selection and optimization and regulatory decisions during all stages of drug development. In general, it is assumed that the E–R relationship is driven by the exposure and, therefore, a positiv…
Jun 18th • 30 mins read

Real-World Evidence: Bridging Gaps in Evidence to Guide Payer Decisions
Real-world evidence (RWE) obtained from an analysis of real-world data (RWD) is a valuable tool that can be used to aid healthcare decision-making. RWD could be derived prospectively or retrospectively from multiple sources, including electronic health records, claims databases, pragmatic trials, or…
Jun 18th • 6 mins read

Comment on: Oncology research in Saudi Arabia over a 10-year period. A synopsis
First, The authors did not comment on the category of research which could be particularly relevant to the local population including screening for early detection, correlative translational research for mechanisms of drug resistance and predictive markers of response, pharmacogenomic-based studies …
Jun 24th • 3 mins read

The regulatory landscape of precision oncology laboratory medicine in the United States - Perspective on the past 5 years and considerations for future regulation
The current state of laboratory test regulation in the United States (US) is complex and the prospect of changes in the current paradigm has been continually on the horizon since 2014. Briefly, clinical laboratories in the US are regulated under the Clinical Laboratory Improvement Amendments of 1988…
May 22nd • 8 mins read

Sponsorship of oncology clinical trials in the United States according to age of eligibility
The sponsor of a clinical trial is the single entity responsible for the overall conduct and oversight of the trial. Sponsors play a critical role in the design and reporting of clinical trials, with most trials sponsored by the pharmaceutical industry, government agencies, or academic institutions.…
Apr 29th • 8 mins read

The Oncology Data Network (ODN): Methodology, Challenges, and Achievements
The Oncology Data Network (ODN) is a cooperative collaborative European data‐sharing platform providing near real‐time information on cancer medicine usage at scale. Its rationale and vision were described in a recent Commentary. Figure 1 summarizes recent advances in oncology and the conse…
May 21st • 8 mins read

Assessment of Clinical Trials Supporting US Food and Drug Administration Approval of Novel Therapeutic Agents, 1995-2017
In the United States, the US Food and Drug Administration (FDA) issues approvals for new drugs and biologics that have demonstrated safety and efficacy in “adequate and well-controlled studies.” Pivotal trials are the most critical of these trials, often identified directly by FDA review…
Apr 21st • 20 mins read

Proportion of Patients in Phase I Oncology Trials Receiving Treatments That Are Ultimately Approved
Phase I oncology trials are designed to evaluate the safety, tolerability, and dosing for new therapeutic strategies. Because they provide access to promising investigational treatments, many patients, oncologists, and policy-makers regard them as a therapeutic option for patients who meet eligibili…
Apr 1st • 14 mins read
Access to Novel Drugs for Non-Small Cell Lung Cancer in Central and Southeastern Europe: A Central European Cooperative Oncology Group Analysis
Lung cancer is the most frequent cause of cancer‐related mortality worldwide, with high incidence and mortality rates in Central and Eastern Europe (CEE). Most patients are diagnosed with advanced disease, resulting in poor survival rates. However, there is a trend toward better outcomes in develo…
Nov 24th • 10 mins read

Publicly accessible evidence of health-related quality of life benefits associated with cancer drugs approved by the European Medicines Agency between 2009 and 2015
Cancer drugs should be clinically meaningful and thus not only prolong but also improve (or at least not worsen) patients' quality of life. Due to the increasing number of cancer drugs in the development pipeline and their rapidly increasing costs, it becomes more and more important to assess the ov…
Feb 23rd • 12 mins read

Patient burden and clinical advances associated with post approval monotherapy cancer drug trials: a retrospective cohort study
After new cancer drugs receive regulatory approval, researchers often pursue trials testing the drug in indications or combinations that would extend the use of the drug beyond the indication for which it was initially approved. Sometimes, such efforts are aimed at obtaining a revision to an Food an…
Feb 17th • 7 mins read
In Vitro-to-In Vivo Extrapolation of Transporter Inhibition Data for Drugs Approved by the US Food and Drug Administration in 2018
The evaluation of the risk of transporter-based drug interactions is now an integral part of the drug development process, supporting the safe use of new treatments in the intended patient populations. A systematic, risk-based, integrated approach, including in vitro, in silico, and i…
Jan 25th • 12 mins read
EHA evaluation of the ESMO-Magnitude of Clinical Benefit Scale version 1.1 (ESMO-MCBS v1.1) for hematological malignancies
In recent years, rapid developments in haematology research resulted in a considerable expansion of treatment options. The development of instruments to measure clinical benefit is essential in the current scenario where increasing numbers of treatments for haematological malignancies (HMs) are beco…
Jan 20th • 20 mins read
Precision medicine for pediatric cancers lags behind that for adult cancers: Citing hurdles in clinical trial recruitment and drug development, researchers are exploring ways to close gaps
Although scientists have made great strides in applying breakthroughs in precision medicine to many adult cancers, the same cannot be said of pediatric diseases. However,researchers, advocates, and lawmakers are hoping to bridge the divide, largely by identifying more opportunities to include childr…
Jan 9th • 2 mins read

Developing a framework to incorporate real-world evidence in cancer drug funding decisions: the Canadian Real-world Evidence for Value of Cancer Drugs (CanREValue) collaboration
New therapies for cancer are becoming increasingly more expensive, challenging the affordability and sustainability of public drug programs in Canada and around the world. While the cost increases of new cancer therapies are evident, the benefits are not always clear. In the Canadian cancer drug fun…
Jan 7th • 8 mins read
Rise of Antibody-Drug Conjugates: The Present and Future
Antibody-drug conjugates (ADCs) are a rapidly emerging class of therapeutic agents that combine the target specificity of a monoclonal antibody (mAb) with the lethality of cytotoxic cellular poison. With ongoing advancements in drug engineering and fresh biologic insight into mechanisms of drug acti…
May 25th • 20 mins read

The pitfalls and promise of liquid biopsies for diagnosing and treating solid tumors in children: a review
The analysis of circulating cell-free nucleic acids is being introduced in several medical fields. In obstetrics, noninvasive prenatal aneuploidy screening for trisomy 21 is well established and widely implemented with high sensitivity and specificity [1]. In transplantation medicine, the amount of …
Jan 3rd • 10 mins read

Real-World Evidence in Oncology: Opportunities and Limitations
Traditionally, randomized controlled clinical trials (RCTs) have been considered the highest level of evidence to define the efficacy of treatments, before their adoption in clinical practice. However, in oncology, like in other fields of medicine, the analysis of real-world evidence (RWE) to answer…
Dec 24th • 8 mins read

Outcome measures for oncology alternative payment models: practical considerations and recommendations
Several alternative payment models (APMs) are being piloted to address affordability, equity and quality-of-care challenges in oncology care. Of these arrangements, the Oncology Care Model (OCM), developed by the Centers for Medicare and Medicaid Innovation (CMMI), is among the most extens…
Dec 1st • 10 mins read

Value assessment of oncology drugs using a weighted criterion-based approach
In recent years, the number of available oncology treatments has significantly increased and these treatments have become more costly, prompting a growing need for sustainable drug funding decision-making processes. In the United States, Canada, and many other countries around the world, cancer drug…
Dec 20th • 15 mins read

Mandatory Research Biopsy Requirements Delay Initiation of Clinical Trials
The number of biomarker directed clinical trials and cancer treatments is increasing (1). This trend is driven by a series of clinical experiences that demonstrate significant improvement in clinical outcomes and progression free survival in many cancers when biomarker directed therapy is employed (…
Oct 18th • 10 mins read
Pivotal Considerations for Optimal Deployment of Healthy Volunteers in Oncology Drug Development
Clinical pharmacology studies (i.e., trials where the primary objectives are traditionally pharmacokinetic (PK)-related) focus on identifying and confirming appropriate dosing in various subsets of the intended patient populations. In most therapeutic areas, these trials are conducted in normal…
Oct 31st • 20 mins read
Publication statuses of clinical trials supporting FDA-approved immune checkpoint inhibitors: a meta-epidemiological investigation
An improved understanding of the biology of cancer has led to remarkable progress in therapeutic approaches. Anticancer agents developed over the last 2 decades utilize multiple mechanisms of action including conventional cytotoxic agents as well as inhibition of oncogenic signalling pathways and an…
Oct 24th • 18 mins read

First person profile: Supriya G. Mohile, MD, MS: A respected geriatric oncologist, Dr. Mohile has conducted innovative research on improving care for older patients with cancer
Beverly Canin, an 85-year-old breast cancer survivor and patient advocate, remembers when she first met Supriya G. Mohile, MD, MS, a geriatric oncologist and the Philip and Marilyn Wehrheim Professor in the division of hematology/oncology at the University of Rochester Medical Center (URMC) in New Y…
Oct 11th • 2 mins read
New Realities of Phase I Clinical Trials in the Era of
The rapid emergence of cancer immunotherapy has been driven by unique development strategies including novel study designs, resulting in new therapeutics being brought to the market with unprecedented speed. For example, development of durvalumab, a human immunoglobulin G1 (IgG1) anti-programmed dea…
Oct 7th • 5 mins read

Liquid biopsy in oncology: a consensus statement of the Spanish Society of Pathology and the Spanish Society of Medical Oncology
By the year 2030, 22.2 million new cases of cancer are expected worldwide: a challenge for cancer patient diagnosis and therapeutic approaches. Despite this increase, patient prognosis has improved with a gradual decrease in cancer-related mortality, reflecting the breakthroughs in early diagnosis a…
Sep 26th • 16 mins read

Prediction of Drug Approval After Phase I Clinical Trials in Oncology: RESOLVED2
Drug development in oncology is a fast-evolving field with numerous challenges. More than 1,000 antineoplastic agents (ANAs) were under investigation in 2018. Oncology had the highest overall attrition rate for US Food and Drug Administration (FDA) approval from phase I (95% between 2006 and 2015), …
Sep 20th • 12 mins read

Association of National Cancer Institute-Sponsored Clinical Trial Network Group Studies With Guideline Care and New Drug Indications
The National Cancer Institute (NCI) Clinical Trial Network (NCTN) groups serve a vital role in identifying new cancer treatments. Unlike pharmaceutical companies—whose primary aims are to develop new drugs and generate profits—the mandate of the NCTN is to serve the community of patients…
Sep 4th • 17 mins read

The rise of oncology biosimilars: from process to promise
Biologics are an increasingly central element of the cancer treatment armamentarium. Unlike traditional pharmaceuticals, which are usually small molecules, biologics are large compounds derived from living organisms, or recombinant proteins produced by cells transformed with the genetic information …
Aug 23rd • 18 mins read
Early-drug development in the era of immuno-oncology: are we ready to face the challenges?
During the past few years we have faced an unprecedented evolution in the design of immunotherapy phase I (Ph1) trials. This change is mainly due to the desire to facilitate patient’s access to drugs with promising activity from early stages of development, and also a consequence of pharmaceut…
Jun 26th • 26 mins read

First person profile: Leslie Bernstein, PhD: An epidemiologist known for her groundbreaking discoveries about breast cancer, Dr. Bernstein continues to push the field forward
When Leslie Bernstein, PhD, joined the faculty of City of Hope at the age of 67, she told the institution's leaders that she would stay for no more than a decade. After all, she had just spent 25 years at the University of Southern California (USC) in Los Angeles, serving as a professor in the …
Aug 12th • 4 mins read

Level of evidence used in recommendations by the National Comprehensive Cancer Network (NCCN) guidelines beyond Food and Drug Administration approvals
The National Comprehensive Cancer Network® (NCCN®) is a not-for-profit alliance of 28 leading cancer centers in the United States devoted to patient care, research, and education. The NCCN mission includes improving the quality, effectiveness, and efficiency of cancer care so that patients c…
Aug 2nd • 8 mins read

Median Survival or Mean Survival: Which Measure Is the Most Appropriate for Patients, Physicians, and Policymakers?
Understanding the efficacy of treatments is crucial for patients, physicians, and policymakers. Median survival, the most common measure used in the outcome reporting of oncology clinical trials, is easy to understand; however, it describes only a single time point. The interpretation of the hazard …
Jul 18th • 15 mins read

Cost per Event Averted in Cancer Trials in the Adjuvant Setting From 2018 to 2022
Globally, the price of cancer drugs is on an upward trajectory, with forecasts of global oncology therapeutic sales reaching $250 billion by 2024. In the US, the Centers for Medicare and Medicaid Services estimates that spending on healthcare will increase from USD 3.6 trillion in 2018 to approximat…
Jun 10th • 30 mins read

Comparison of Long-term Survival Benefits in Trials of Immune Checkpoint Inhibitor vs Non-Immune Checkpoint Inhibitor Anticancer Agents Using ASCO Value Framework and ESMO Magnitude of Clinical Benefit Scale
Conventional non–immune checkpoint inhibitor (ICI) anticancer agents typically improve patients’ overall survival (OS) and progression-free survival (PFS). However, long-term survival benefits are limited by acquired biological resistance. In contrast to non-ICI agents, ICI agents h…
Jul 10th • 12 mins read

Clinical benefit of cancer drugs approved in Switzerland 2010–2019
Over time, the number of cancer drugs approved by the Food and Drug Administration (FDA) has increased. This has been attributed to advances in cancer drug research but also to faster approvals which are often based on preliminary data and intermediate primary endpoints instead of overall survival (…
Jun 10th • 35 mins read

A Comprehensive Comparison of Additional Benefit Assessment Methods Applied by Institute for Quality and Efficiency in Health Care and European Society for Medical Oncology for Time-to-Event Endpoints After Significant Phase III Trials—A Simulation Study
Pharmaceutical companies search for new drugs against specific diseases, for example cancer, which have to be investigated with respect to their quality, safety, and efficacy, preventing a nonbeneficial drug from getting on the market. Most efficacy endpoints in cancer trials are time-to-event endpo…
Jun 28th • 30 mins read

Uptake of Oncology Biosimilars: Managed Care Strategies to Improve Value-Based Care Systems
In recent years, the cost of cancer care associated with biologics in the United States has increased at an alarming rate, causing some patients to be priced out of treatment. Biosimilars present important opportunities to lower healthcare costs and expand access to care for oncology patients. Oncol…
Jul 7th • 25 mins read
Audit of Data Sharing by Pharmaceutical Companies for Anticancer Medicines Approved by the US Food and Drug Administration
Decisions by regulators and clinicians on whether to approve and use new medications are typically based on findings from pivotal clinical trials. For most newer medicines, an industry sponsor drives the early generation of the evidence base supporting the medicine, but this process requires access …
Jul 28th • 20 mins read

Overall Survival Benefits of Cancer Drugs Approved in China From 2005 to 2020
The primary goal of cancer drug therapy is to prolong life or improve quality of life. Overall survival (OS) is the most reliable clinical trial endpoint to inform regulatory approvals of new cancer drugs. However, most cancer drugs approved in the US and Europe lack evidence of OS benefit. For inst…
Aug 10th • 30 mins read

Recent Trends in Medicaid Spending and Use of Drugs With US Food and Drug Administration Accelerated Approval
State Medicaid programs have expressed increasing concern about rising prescription drug spending. Some states have expressed concern about constraints on the strategies available to them for cost containment under the Centers for Medicare & Medicaid Services (CMS) Medicaid Drug Rebate Program, …
Oct 8th • 25 mins read

How do cancer clinicians perceive real-world data and the evidence derived therefrom? Findings from an international survey of the European Organisation for Research and Treatment of Cancer
Before a novel medicine can enter the market, its manufacturer must obtain marketing authorization from the competent regulatory authorities. To satisfy the stringent evidentiary requirements imposed by regulators such as the European Medicines Agency (EMA), the US Food and Drug Administration (FDA)…
Aug 1st • 45 mins read

FDA validation of surrogate endpoints in oncology: 2005–2022
Approximately two-thirds of cancer drugs receive US Food and Drug Administration FDA marketing authorization based upon improvement in a surrogate endpoint, such as tumor shrinkage or progression-free survival. While surrogates may allow for earlier entry of drugs into the market, they are not alway…
Dec 1st • 20 mins read

Research Highlights In The News From ESMO
In September 2022, the ESMO 2022 Congress was held in Paris, marking the return of a highly anticipated annual in-person event for the oncology community. The conference attracted 29,000 delegates, with approximately 5,000 joining the event online. The congress featured a diverse program, carefully…
Sep 30th • 10 mins read

Oncology biosimilars: New developments and future directions
Biologicals have become an integral part of cancer treatment both as therapeutic agents and as supportive care agents. It is important to know that biologics are large, complex molecular entities requiring extensive immunogenicity testing and pharmacovigilance strategies to ensure no immune response…
Nov 25th • 30 mins read

Association Between US Drug Price and Measures of Efficacy for Oncology Drugs Approved by the US Food and Drug Administration From 2015 to 2020
The US has worse cancer-related outcomes than other high-income countries while bearing the highest cost of cancer care in the world. A reason for increasing cost may be improved efficacy of expensive novel agents. One study found an association between measures of benefit and price, although the st…
Oct 31st • 10 mins read
Identification of Barriers Preventing Biosimiliar Oncology Medication Adoption
Biosimilars are FDA-approved medications that are biological in type and contain a very similar substance that has been previously approved by the FDA. Similar to other generic medications, biosimilars offer an opportunity for lower healthcare costs due to intrinsic price competition with the origin…
Oct 27th • 30 mins read

Association between control group therapy and magnitude of clinical benefit of cancer drugs
Several oncology societies have developed tools to quantify the magnitude of clinical benefit of drugs for the treatment of solid tumors. These include the American Society of Clinical Oncology Value Framework (ASCO-VF), the European Society for Medical Oncology Magnitude of Clinical Benefit Scale (…
Dec 9th • 20 mins read

Methodological and reporting standards for quality-of-life data eligible for European Society for Medical Oncology-Magnitude of Clinical Benefit Scale
In 2015, the European Society for Medical Oncology (ESMO) introduced the ESMO-Magnitude of Clinical Benefit Scale (ESMO-MCBS), a validated tool to stratify the magnitude of clinical benefit derived from therapeutic approaches in a standardized way. With the rapid increase in new treatment options an…
Apr 1st • 30 mins read

Biosimilars in Oncology: Latest Trends and Regulatory Status
Biological therapeutic agents, commonly known as biologics, encompass a diverse range of substances produced by cells or living organisms through various biological processes, such as controlled gene expression, antibody technologies, and recombinant DNA technologies. Over recent decades, biologics …
Dec 5th • 20 mins read

Towards a novel approach guiding the decision-making process for anticancer treatment in patients with advanced cancer: framework for systemic anticancer treatment with palliative intent
Oncologists typically base treatment recommendations on survival, tumor control, and toxicity data from high-quality clinical trials.¹ In patients with resistant tumors and in unfit patients, anticancer treatment is often applied even without sufficient supporting evidence. Many patients may fa…
Jun 1st • 25 mins read

Patient involvement: A must-have in medicine development, but is it being overlooked in a cost-constrained environment?
The life sciences industry is in a constant state of advancement, bringing more and more groundbreaking medicines, cutting-edge technologies, and innovative solutions to market. Amidst these rapid changes, patients remain at the heart of these scientific developments. In recent decades, the l…
May 9th • 5 mins read

Medical affairs: The power behind redefining commercialization
In March 2024, Envision Pharma Group held a Medical Affairs Professional Society (MAPS) Americas roundtable with leaders discussing the role of science in commercialization strategies and the importance of integrating robust scientific understanding into the commercial journey. Envision revisite…
Aug 2nd • 3 mins read

Empowering people to drive medical affairs performance with AI
Artificial intelligence (AI) is transforming the pharmaceutical industry and continues to be a key topic of discussion in the field. As medical affairs grow more complex, AI can significantly reduce the time required to analyze and extract valuable insights, thereby accelerating the generation of ac…
Aug 14th • 5 mins read
Exposure to US Cancer Drugs With Lack of Confirmed Benefit After US Food and Drug Administration Accelerated Approval
Between 2009 and 2022, the US Food and Drug Administration (FDA) approved 48 drugs for 66 oncology-related indications under the Accelerated Approval (AA) program. Indications granted AA based on surrogate end points are subsequently required to confirm clinical benefit. Since 2009, 15 indications (…
Dec 8th • 2 mins read

The Value of Pharmaceutical Industry-Sponsored Patient Registries in Oncology Clinical Research
In May 2019, the US Food and Drug Administration (FDA) released the Framework for FDA’s Real-World Evidence (RWE) Program, a draft guidance to evaluate the potential use of real-world data (RWD) in facilitating regulatory decisions. The guidance was a clear indication of the increasi…
Jun 7th • 8 mins read
Value assessment of PD-1/PD-L1 inhibitors in the treatment of esophageal and gastrointestinal cancers
According to GLOBOCAN data, colon cancer, gastric cancer (GC), rectal cancer and esophageal cancer (EC) are among the top 10 cancers in terms of incidence, and digestive system cancers have become one of the most serious disease burdens. In recent years, the use of programmed cell death 1 …
Apr 21st • 13 mins read

Single-arm trials supporting the approval of anticancer medicinal products in the European Union: contextualization of trial results and observed clinical benefit
Randomized controlled trials (RCTs) are referred to as the ‘gold standard’ in testing medicinal products. These trials have several advantages over clinical trials with other designs due to their design features. For example, randomization facilitates subjects in the experimental and con…
Apr 11th • 14 mins read

Defining the role of real-world data in cancer clinical research: The position of the
The emergence of the precision medicine paradigm in oncology has led to increasing interest in the integration of real-world data (RWD) into cancer clinical research. As sources of real-world evidence (RWE), such data could potentially help address the uncertainties that surround the adoption of nov…
Mar 20th • 10 mins read

Evaluating External Validity of Oncology Biosimilar Safety Studies
Biologics are the fastest growing medication class in the US and account for an increasing portion of health care costs. In the US, the Biologics Price Competition and Innovation Act of 2009 created an abbreviated approval pathway to expand access to safe and effective biological products. Pivo…
Apr 6th • 2 mins read

Preliminary Attainability Assessment of Real-World Data for Answering Major Clinical Research Questions in Breast Cancer Brain Metastasis: Framework Development and Validation Study
Brain metastasis (BM) is a major cause of mortality in patients with breast cancer, and it increases the difficulty of treatment. Advancements in treatment and the development of brain imaging technology have increased the survival of patients with metastatic breast cancer, leading to an increased i…
Oct 9th • 4 mins read

The correlation between the costs and clinical benefits of PD-1/PD-L1 inhibitors in malignant tumors: An evaluation based on ASCO and ESMO frameworks
Survival benefits of patients with a malignant tumor have been improved significantly over the years, partially attributed to the employment of novel anti-cancer therapies. Recent success in immunotherapy propels cancer treatment to an exciting new era after traditional chemotherapy and targete…
Feb 23rd • 9 mins read

Value assessment of NMPA-approved new cancer drugs for solid cancer in China, 2016-2020
Innovations in cancer therapy, particularly the influx of new drugs have yielded high expectations of transform treatment of the disease from all healthcare stakeholders. Nevertheless, dramatic rise in drug costs has recently highlighted a vigorous debate over whether cancer drugs prices, especially…
Feb 24th • 8 mins read
Early phase clinical trial played a critical role in the Food and Drug Administration-approved indications for targeted anticancer drugs: a cross-sectional study from 2012 to 2021
With the development of precision oncology, molecular targeted anticancer drug has become the most attractive research field in new drug development A lot of time, money and resources have been invested in developing novel targeted anticancer drugs. More and more blockbuster drugs have been approved…
Mar 9th • 10 mins read

Off-label despite high-level evidence: a clinical practice review of commonly used off-patent cancer medicines
Cancer is among the leading causes of death and an ongoing challenge for health care systems worldwide. During the last decade, a plethora of new medicines have been approved by regulatory bodies for the treatment of neoplastic diseases and in most countries, medicines are reimbursed according to th…
Nov 14th • 21 mins read

US Government Payer-Funded Trials to Address Oncology's Drug-Dosing Conundrum: A Congressional Call to Action?
Since the mid-20th century, cancer drug development has been guided by the assumption that more is better.1 Early phase clinical trials of new drugs therefore sought the maximum tolerated dose, which would then be compared with the standard of care in a randomized trial. In nearly all other are…
Feb 13th • 5 mins read

What is the weight of expectation bias in oncology trials?
In biomedical research, expectation bias, also referred to as Rosenthal effect, is the distorting effect on the results of an experiment caused by the expectation that the investigator, or the patient, has about the results themselves. The classical way to reduce the influence of this bias on t…
Feb 11th • 3 mins read
Factors associated with the uptake of biosimilars for breast cancer treatment from the perspectives of physicians and patients-Evidence from China
Unlike small-molecular chemical products with well-specified structures, biological agents are macromolecular substances with complex structures and manufacturing processes and are usually highly priced . Taking cancer medicines as an example, biological agents are increasingly highly pric…
Jan 12th • 13 mins read

Twelve ESMO Congress 2022 breakthroughs: practicing oncologists' perceptions and potential application on presented data
During the European Society for Medical Oncology (ESMO) Congress 2022, outcome data of a great number of trials were presented, some of which are expected to change daily clinical practice. For the attending medical oncologist, it is important to structure this vast amount of data in a way that faci…
Jan 10th • 12 mins read

Medical Oncologists’ Knowledge and Perspectives on the Use of Biosimilars in the United States
To assist in cost containment across medicine, the Biologics Price Competition and Innovation Act of 2009 was enacted to create an abbreviated approval pathway for biological products that are very similar to approved biologics. Where patents for successful biologics are expiring, biosimil…
Jan 9th • 9 mins read
Application of Value Framework to Phase III Trials of Immune Checkpoint Inhibitors in Esophageal and Gastric Cancer
Advanced and metastatic esophageal and gastric cancer portends poor prognosis with limited durable treatment options. In 2020, GLOBOCAN estimated approximately 544 076 deaths globally from esophageal cancer, which is one of every 18 cancer-related deaths, and 768 793 deaths from s…
Jan 13th • 8 mins read

Embracing Project Optimus: Can we Leverage Evolutionary Theory to Optimize Dosing in Oncology?
Demonstration of superior clinical efficacy is a major barrier to the approval of investigational oncology drugs. For indications with limited treatment options, sponsors frequently conduct efficacy trials under expedited timelines in hopes of making their therapy available to patients in need as qu…
Sep 2nd • 10 mins read

Are the chronological age cutoffs used in clinical oncology guidelines biologically meaningful?
Cancer is predominantly a disease of ageing: the incidence of many epithelial tumours increases with age. With a peak incidence at around 70 years of age, breast cancer is no exception. Moreover, more than one-third of all breast tumours are diagnosed in patients over this age. In older patient…
Dec 1st • 4 mins read

The challenges and opportunities in using real-world data to drive advances in healthcare in East Asia: expert panel recommendations
Real-world data (RWD) relating to patient health status and/or the delivery of healthcare can be broadly defined as data that are collected outside of conventional clinical trials. These data are derived from a variety of sources, including electronic medical records (EMRs), insurance claims and bil…
Jun 28th • 13 mins read

Rationale, Strengths, and Limitations of Real-World Evidence in Oncology: A Canadian Review and Perspective
Randomized controlled trials (RCTs) remain the gold standard for evaluating the efficacy of cancer therapies. Their design, however, limits the amount and type of information they can. For clinicians, researchers, and regulators to develop a more complete and in-depth understanding of therapeutic ag…
Apr 26th • 9 mins read
Building a Healthcare Alliance for Resourceful Medicine Offensive Against Neoplasms in Hematology Added Value Framework for Hematologic Malignancies: A Comparative Analysis of Existing Tools
In 2012, costs associated with hematologic malignancies (HMs) in the European Union (EU) reached €12 billion accounting for 8% of total EU cancer costs (€143 billion). This consisted of direct healthcare costs of €7·3 billion, productivity losses of €3.6 billion, and infor…
May 17th • 12 mins read

External control arms in oncology: current use and future directions
The control arm of a randomized clinical trial plays a fundamental role in estimating the efficacy and safety of an investigational therapy. Concurrently randomized control arms allow for an understanding of the temporally relevant factors associated with the natural history of the disease, par…
Jan 9th • 9 mins read

CheckMate-067: Raising the Bar for the Next Decade in Oncology
Few effective systemic therapies were available for patients with advanced melanoma until recently, although durable complete responses were achieved in a small percentage of patients treated with high-dose interleukin-2. In 2015, results from the phase III CheckMate-067 trial led to US Food and Dru…
Dec 2nd • 2 mins read

Report from American Society of Clinical Oncology Symposium 2020 and American Society of Clinical Oncology Gastrointestinal Cancer Symposium 2021
Patients with cancer need timely access to drugs that meaningfully improve how they feel, function, or survive. However, measuring clinical endpoints such as improvement in overall survival can take time, and patients may be willing to tolerate uncertainty about such benefits in exchange for early a…
Aug 4th • 13 mins read

Real-World Evidence in Support of Oncology Product Registration: A Systematic Review of New Drug Application and Biologics License Application
Regulatory real-world evidence (RWE) has garnered great interest since the passing of the 21st Century Cures Act in 2016, which aimed to accelerate medical product development by supporting approval of new indications for current therapies or satisfying post-approval requirements (1–3).  …
Jan 1st • 12 mins read

Estimated Medicare Spending on Cancer Drug Indications With a Confirmed Lack of Clinical Benefit After US Food and Drug Administration Accelerated
The accelerated approval (AA) pathway expedites the market entry of new drugs by allowing the US Food and Drug Administration (FDA) to grant approval using surrogate end points (eg, progression-free survival) that are “reasonably likely” to predict clinical benefit (eg, overall survival …
Oct 18th • 5 mins read

Regulatory and clinical consequences of negative confirmatory trials of accelerated approval cancer drugs: retrospective observational study
Patients with cancer need timely access to drugs that meaningfully improve how they feel, function, or survive. However, measuring clinical endpoints such as improvement in overall survival can take time, and patients may be willing to tolerate uncertainty about such benefits in exchange for early a…
Aug 4th • 12 mins read

The Inclusion of Women in Global Oncology Drug Trials Over the Past 20 Years
Thirty years have passed since the enactment of the National Institutes of Health (NIH) Revitalization Act, which encouraged NIH-funded investigators to include adequate numbers of women in clinical studies. Since then, there have been important steps taken to ensure better representation of women a…
Aug 26th • 2 mins read
Implications of Research Biopsies in Clinical Trials
Biospecimens are invaluable lynchpins in translational oncology, both to engender new discoveries and to assess the efficacy of new treatment paradigms. The collection and use of such specimens rely upon the patients and subjects who entrust clinicians with their bodies. This is especially true …
Aug 31st • 2 mins read
Canadian Regulatory and Health Technology Assessment for Malignant Hematology and Oncology Indications Compared With the US Food and Drug Administration Accelerated Approval Program
For serious or life-threatening diseases such as cancer, there is tension between standard regulatory approval processes and patients’ demand for early access to treatments. To address this issue, the US Food and Drug Administration (FDA) initiated the Accelerated Approval (AA) expedited pathw…
Jun 5th • 6 mins read

Payer perceptions of the use of real-world evidence in oncology-based decision making
Innovative therapies for cancers are being introduced with growing frequency. Some of these therapies, especially those in areas with high unmet needs and limited patient populations, receive accelerated regulatory approvals, sometimes based on Phase II studies only or single-arm trials. In 2012, th…
Aug 1st • 12 mins read

Assessment of gender representation in clinical trials leading to FDA approval for oncology therapeutics between 2014 and 2019: A systematic review-based cohort study
Gender disparities in health care have influenced processes for appropriate diagnoses and treatment of many health conditions. Among patients with cancer, these epidemiological variations have driven advancements to alleviate gender-specific differences in cancer susceptibility and mortality. Clinic…
Jun 23rd • 8 mins read

Use of real-world evidence for oncology clinical decision making in emerging economies
Real-world evidence (RWE) can provide insights into patient profiles, disease detection, treatment choice, dosing strategies, treatment sequencing, adverse event management and financial toxicity associated with oncology treatment. However, the full potential of RWE is untapped in emerging economies…
May 5th • 12 mins read

Patient Participation in Clinical Trials of Oncology Drugs and Biologics Preceding Approval by the US Food and Drug Administration
Recent studies estimate the median cost of bringing a new drug to market at $985.3 million for all therapeutic agents and $793.4 million for oncology drugs. For every 100 drugs entered into phase 1 clinical testing in oncology, fewer than 10 will ultimately receive regulatory approval. A minority of…
May 18th • 5 mins read

Patient-Reported Outcomes in Pediatric Cancer Registration Trials: A US Food and Drug Administration Perspective
The 21st Century Cures Act directed the US Food and Drug Administration (FDA) to systematically incorporate patients’ experiences, needs, perspectives, and priorities into drug development and evaluation. Patients are experts in their disease because of their lived experience with its symptoms…
Apr 30th • 12 mins read
Model-Informed Therapeutic Dose Optimization Strategies for Antibody-Drug Conjugates in Oncology: What Can We Learn From US Food and Drug Administration-Approved Antibody-Drug Conjugates?
Antibody–drug conjugates (ADCs) combine the specificity of an antibody with the cytotoxicity of a chemical agent. They represent a rapidly evolving area of oncology drug development and hold significant promise. There are currently nine ADCs on the market, more than half of which gained US Foo…
Apr 26th • 15 mins read

Oncology approvals in 2020: a year of firsts in the midst of a pandemic
To limit potential exposure to SARS-CoV-2, in 2020 the FDA made several approvals aimed at reducing the frequency of interactions between patients with cancer and their health-care providers. These approaches included increasing dosing intervals, such as that for pembrolizumab (which was extended fr…
Jan 29th • 4 mins read
FDA Oncology Center of Excellence Project Renewal: Engaging the Oncology Community to Update Product Labeling for Older Oncology Drugs
The FDA-approved product labeling is a summary of information verified during FDA's independent, unbiased review of pharmaceutical quality, nonclinical and clinical pharmacology, and clinical study data. In 2006, the Physician Labeling Rule (PLR) was published, creating a consistent content and…
Feb 15th • 13 mins read
Inadequate and delayed characterization of cutaneous reactions for US Food and Drug Administration-approved oncologic drugs from 2011-2020 leading to medication discontinuation
Cutaneous reactions are common adverse events (AE) in oncologic drug clinical trials and a frequent reason for discontinuation. Although properly diagnosing cutaneous AEs and keeping patients on oncologic therapy is increasingly appreciated, many cutaneous AEs are not detailed in subsequent studies …
Oct 23rd • 4 mins read

Use of real-world evidence in cancer drug funding decisions in Canada: a qualitative study of stakeholders’ perspectives
The costs of cancer drugs are rising rapidly. Although some new drugs provide substantial therapeutic improvements, others confer only marginal survival benefits or improve only quality of life. Assessment of the overall costs and benefits of cancer drugs is essential for resource allocation. Drug f…
Nov 4th • 12 mins read

The Evidence REVEAL Study: Exploring the Use of Real-World Evidence and Complex Clinical Trial Design by the European Pharmaceutical Industry
The rapid evolution of science and technology allows innovative approaches to generate new types of evidence about the effectiveness of medical product development so as to speed up patients’ access to better diagnostics and treatment. Our study explored how two emerging approaches, the use of…
Nov 20th • 10 mins read

Clinical Trial Evidence Supporting US Food and Drug Administration Approval of Novel Cancer Therapies Between 2000 and 2016
Cancer research is characterized by the perceived urgency to develop novel drugs that may improve patients’ survival and quality of life. Before patients have access to novel therapies, the available evidence on benefits and harms from clinical trials is assessed by authoritative institutions,…
Nov 10th • 6 mins read

Potential Cost Implications for All US Food and Drug Administration Oncology Drug Approvals in 2018
Rising drug prices and national expenditures are receiving increasing attention after 2018 yielded a record 59 novel drug approvals by the US Food and Drug Administration (FDA). In the US, cancer drug spending has grown 12% to 15% annually and is projected to continue on this trajectory in the comin…
Aug 31st • 7 mins read

Use of Real-World Evidence to Support FDA Approval of Oncology Drugs
In December 2016, the 21st Century Cures Act (“Cures Act”) was signed into United States (US) law. The Cures Act, designed to improve the efficiency and speed of new medical product development and regulatory approval, mandated that the Food and Drug Administration (FDA) establish a prog…
Sep 14th • 16 mins read

Approval of Cancer Drugs With Uncertain Therapeutic Value: A Comparison of Regulatory Decisions in Europe and the United States
In the United States (US) and the European Union (EU), regulatory agencies—the US Food and Drug Administration (FDA) and the European Medicines Agency (EMA), respectively—are responsible for assessing the benefit-risk profile of new drugs before they enter the market. The mandate of thes…
Oct 6th • 48 mins read

Discordance Between Child-Pugh and National Cancer Institute Classifications for Hepatic Dysfunction: Implications on Dosing Recommendations for Oncology Compounds
Efficacy and safety of new anticancer agents are often established in clinical studies that exclude patients with advanced degrees of organ impairment. Therefore, dedicated hepatic impairment (HI) studies are conducted to compare drug exposure in subjects with varying degrees of liver dysfunction wi…
Jul 20th • 18 mins read
Mechanistic Quantitative Pharmacology Strategies for the Early Clinical Development of Bispecific Antibodies in Oncology
Cancer is a complex, multifactorial disease. Crosstalk between signaling cascades and multiple mediators of tumor survival and immune evasion exist. Genetic alterations lead to heterogeneity in tumor cell antigen expression within and between patients. Acquisition of resistance to therapy is as…
Jun 24th • 18 mins read
Sponsorship of oncology clinical trials in the United States according to age of eligibility
The sponsor of a clinical trial is the single entity responsible for the overall conduct and oversight of the trial. Sponsors play a critical role in the design and reporting of clinical trials, with most trials sponsored by the pharmaceutical industry, government agencies, or academic institutions.…
Apr 29th • 6 mins read

FDA Acceptance of Surrogate End Points for Cancer Drug Approval: 1992-2019
The US Food and Drug Administration (FDA) approves cancer drugs based on direct measures of patient benefit—such as overall survival (OS) or quality of life—or surrogate measures, such as change in biomarker level or tumor size on imaging studies. Surrogate end points often have weak or …
Mar 9th • 4 mins read

The Use of Not-Negative Conclusions to Describe Results of Formally Negative Trials Presented at Oncology Meetings
Presentations at medical meetings have great resonance within the scientific community, especially following the diffusion of social media. Oral presentations are not subject to peer review, and some authors’ conclusions may not be completely justified by the results. This is particularly crit…
Feb 13th • 2 mins read

Biased by design? Clinical trials and patient benefit in oncology
A recent study by Naci et al. examined the design characteristics, risks of bias and reporting adequacies of pivotal randomized controlled trials of cancer drugs approved by the EMA in the period 2014–2016. During this period, 32 new cancer drugs were approved by the EMA on…
Nov 27th • 3 mins read

Pediatric Trials for Cancer Therapies With Targets Potentially Relevant to Pediatric Cancers
In 2017, Congress enacted the Research to Accelerate Cures and Equity (RACE) for Children Act to stimulate the development of targeted therapies for pediatric cancers (1). The RACE Act closed a legislative gap that had exempted cancer drugs from mandatory pediatric studies. Specifically, the RACE A…
Oct 29th • 10 mins read

Defining a Clinically Meaningful Benefit in Cancer Clinical Trials: From the Perspectives of the Clinical Trialist, Patient, and Society
In an era of value-based medicine, it is increasingly important to design clinical trials of new therapies that not only show statistically significant benefits compared with current standard therapies but also yield an adequate magnitude of benefit to be deemed “clinically meaningful.” …
Jun 27th • 2 mins read
Dose Finding in the Clinical Development of 60 US Food and Drug Administration-Approved Drugs Compared With Learning vs. Confirming Recommendations
This review characterizes clinical development that supported the label dose in 60 drug indications recently approved by the US Food and Drug Administration. With Lewis B. Sheiner's Learning vs. Confirming clinical drug development paradigm as a reference point, the clinical developme…
Jun 29th • 15 mins read

Addressing oncologists' gaps in the use of biosimilar products
The availability of biosimilar products may improve access to healthcare by increasing the number of therapeutic options available at potentially lower costs. As of April 2019, 18 such biological products had been approved by the FDA, including 4 biosimilars for trastuzumab, 3 each for inflixim…
Jun 19th • 5 mins read
The evolving landscape of precision medicine in primary liver cancer
The incidence of primary liver cancer (PLC) is rising faster than any other malignancy in the USA and is estimated to result in over 31,000 deaths in 2019. PLC poses a unique challenge in that the majority of patients suffer from both their malignancy and underlying liver damage which is the in…
Mar 29th • 3 mins read
Assessing cardiac safety in oncology drug development
The Cardiac Safety Research Consortium (CSRC; www.cardiac-safety.org) held a Think Tank on “Detection, Assessment, and Risk Mitigation of Cardiac Safety Signals in Oncology Drug Development” on October 24-25, 2017, at the Food and Drug Administration (FDA) headquarters in Silver Spr…
Jun 12th • 12 mins read

Recent eUpdates to the ESMO Clinical Practice Guidelines on hepatocellular carcinoma, cancer of the pancreas, soft tissue and visceral sarcomas, cancer of the prostate and gastric cancer
Following the results of three studies, the management of advanced disease, systemic therapies for advanced hepatocellular carcinoma (HCC)-targeted first-line and second-line therapies have been updated. Lenvatinib received European Medicines Agency (EMA) approval in late 2018 and is associated with…
Jun 6th • 2 mins read

An Overview of Cancer Drugs Approved by the US Food and Drug Administration Based on the Surrogate End Point of Response Rate
The US Food and Drug Administration (FDA) approves cancer drugs based on (1) overall survival (OS) or patient reported outcomes, (2) progression-free survival, the time until cancer recurs or worsens, or (3) response rate (RR), the percent of patients experiencing tumor shrinkage. Response rate and …
May 28th • 5 mins read
Quantitative Mechanistic Modeling in Support of Pharmacological Therapeutics Development in Immuno-Oncology
Immunotherapy of cancer has had a long history of development, starting from pioneering efforts in using coley toxins to treat patients—a therapeutic approach named after Dr. William Coley. Even though these earlier efforts never turned into a standard treatment, further investigations on the …
Apr 30th • 12 mins read

Assessment of Whether the American Society of Clinical Oncology's Value Framework and the European Society for Medical Oncology's Magnitude of Clinical Benefit
The American Society of Clinical Oncology (ASCO) and the European Society for Medical Oncology (ESMO) have independently published value frameworks that allow for the systematic assessment of clinical benefit of anticancer drugs with the aim of establishing the value of these therapies. Both framewo…
May 16th • 15 mins read
Association of Industry and Academic Sponsorship With Negative Phase 3 Oncology Trials and Reported Outcomes on Participant Survival: A Pooled Analysis
Phase 3 randomized clinical trials (RCTs) are the final barrier in establishing the efficacy of cancer drugs. One study estimates that only 3.4% of cancer drugs being evaluated in phase 1 trials are approved by the US Food and Drug Administration, with most (35.5%) failing in the phase 3 stage when …
May 10th • 8 mins read
Estimation of the Percentage of US Patients With Cancer Who Are Eligible for and Respond to Checkpoint Inhibitor Immunotherapy Drugs
Cancer checkpoint inhibitors have received considerable and broad interest because of their ability to generate durable responses in many hitherto intractable malignant tumors and for improvements in overall survival in several randomized trials. These promising drugs, and their underlying prec…
May 3rd • 10 mins read

Analysis of Control Arm Quality in Randomized Clinical Trials Leading to Anticancer Drug Approval by the US Food and Drug Administration
Most randomized clinical trials (RCTs) of novel anticancer drugs leading to marketing authorization by the US Food and Drug Administration (FDA) are designed, conducted, and funded by the biopharmaceutical industry. One potential concern with these studies is that in an effort to increase the likeli…
May 2nd • 15 mins read
Cancer drug development: The missing links
Cancer is a group of more than 200 neoplastic diseases, caused by diverse deregulated cell signaling cascades. It represents the leading causes of morbidity as well as mortality across the globe and over the coming two decades, its incidence is predicted to increase by approximately 70% Among a…
Apr 18th • 20 mins read

Cancer experts point to new advances in research and treatment: A recent report by the American Association for Cancer Research highlights unprecedented successes as well as ongoing challenges in the cancer field
Within the last few years, cancer researchers have rapidly gained ground in several key areas, and their progress is visible in a wave of new therapeutics. Twenty-two new treatments were approved by the US Food and Drug Administration (FDA) for various types of cancer between August 1, 2017, and Jul…
Apr 1st • 2 mins read

Biosimilars: what the oncologist should know
An increasingly greater proportion of life-saving therapies now include complex biologics. More and more cancer therapeutics and medications used for inflammatory diseases are counted among this growing class of agents. However, biologics can be expensive and as originator biologics begin to lo…
Dec 18th • 25 mins read

Timing of first-in-child trials of FDA-approved oncology drugs
Progress in treating pediatric malignancies over the last forty years has largely been driven by advances in conventional cytotoxic chemotherapy regimens, surgery and radiation. While overall survival for children with cancer now exceeds 80%, outcomes remain poor for many patients.&nb…
Mar 18th • 10 mins read

Estimation of Study Time Reduction Using Surrogate End Points Rather Than Overall Survival in Oncology Clinical Trials
Overall survival (OS) and validated patient-reported outcomes (PROs) are clinical end points in oncology clinical trials that are intrinsically meaningful to patients. The US Food and Drug Administration (FDA) has the ability to approve cancer treatments on the basis of surrogate end points, such as…
Apr 1st • 10 mins read
Representation of Patients With Cardiovascular Disease in Pivotal Cancer Clinical Trials
Cardiovascular disease (CVD) is increasingly common among new cancer patients, with a reported prevalence near 30% at diagnosis. Cancer patients with concurrent CVD have poorer outcomes than those without CVD. Despite the high prevalence of CVD and its prognostic significance, there is limited…
Mar 18th • 3 mins read
Anticancer drugs approved by the Food and Drug Administration for gastrointestinal malignancies: Clinical benefit and price considerations
By 2020, the cost of cancer care in the United States is expected to reach $158 billion US dollars.1 Pharmaceutical drugs are the fastest growing aspect of US health care spending, with oncology treatments amounting to nearly $37.8 billion in 2015. Over the past decade, the average monthly…
Mar 7th • 8 mins read

Value assessment frameworks in oncology: championing concordance through shared standards
As rapidly evolving tools under the constant rigorous review and development by both ASCO and ESMO’s expert task forces, the ASCO Value Framework Net Health Benefit Score (ASCO-NHB) and the ESMO Magnitude of Clinical Benefit Scale (ESMO-MCBS) were both launched in 2015 to assess the relative b…
Feb 18th • 1 min read
The Case for Real-world Evidence in the Future of Clinical Research on Chronic Myeloid Leukemia
Conventional randomized controlled trials (RCTs) are rigorous scientific examinations of the tolerability and efficacy of potential new therapies. RCTs define precise clinical end points, maximize adherence, minimize variance of clinical conditions, and are limited to relatively homogeneous pa…
Jan 29th • 10 mins read
Overview of Oncology and Hematology Drug Approvals at US Food and Drug Administration Between 2008 and 2016
We previously reviewed the oncology products approved by the FDA between July 2005 and December 2007 (1). Since 2007, however, a number of changes have taken place to accommodate a dramatic increase in applications in oncology. The Office of Oncology Drug Products (OODP), created in July 2005 to ove…
Aug 4th • 15 mins read

Opportunities and challenges in biosimilar uptake in oncology
There are now 10 approved biosimilars in the United States, including 3 oncology drugs, and at least 16 others in late-stage development. The introduction of competition into the biologic space launches a new era inthe treatment of cancer, possibly increasing access to the extremely c…
Jun 26th • 7 mins read

Cancer research in the United States: A critical review of current status and proposal for alternative models
Cancer research in the United States has undergone seismic changes in the past 6 decades. In the early phases, before the second half of the 20th century, cancer laboratory research focused on tissue histology and animal xenograft models, with surgery and radiotherapy being the mainstays of tre…
May 14th • 10 mins read
Reverse Translation of US Food and Drug Administration Reviews of Oncology New Molecular Entities Approved in 2011-2017: Lessons Learned for Anticancer Drug Development
We conducted a comprehensive analysis of clinical pharmacology evaluations in initial submissions of 56 oncology new molecular entities approved by the US Food and Drug Administration between January 2011 and April 2017. Results from studies evaluating food effect, QTc prolongation, drug-drug intera…
Mar 11th • 15 mins read
Challenges and Opportunities in Dose Finding in Oncology and Immuno-oncology
Today's cancer therapy has made substantial progress. Recent success in immuno‐oncology (IO) and combination therapies provide opportunities to enhance activity in the broader population with less drug resistance. However, these advances also raise challenges in oncology drug development. At …
Jul 11th • 10 mins read

Evolving Landscape of US Food and Drug Administration Drug Approval in the Era of Precision Oncology: Finding the Right Balance Between Access and Safety
Since 1992, the US Food and Drug Administration (FDA) has implemented several programs for streamlined review and approval of agents that treat serious or life-threatening conditions, including the Accelerated Approval Pathway as well as designations for Priority-Review, Fast-Track, and Breakthrough…
Jun 20th • 3 mins read

The FDA Oncology Center of Excellence and Precision Medicine
In January 2017, the U.S. Food and Drug Administration (FDA) formally established the Oncology Center of Excellence (OCE) to streamline the development of cancer therapies by uniting experts from FDA product centers to conduct expedited review of drugs, biologics, and devices. In May 2017,…
Nov 6th • 3 mins read

Streamlining Adverse Events Reporting in Oncology: An American Society of Clinical Oncology Research Statement
Monitoring patient safety during clinical trials is critical to protecting research participants from preventable harms as well as protecting patients who will ultimately be treated with an intervention. Under the current regulatory system for investigational new drugs (INDs), sponsors are required …
Feb 20th • 3 mins read

Magnitude of Clinical Benefit of Cancer Drugs Approved by the US Food and Drug Administration
The US Food and Drug Administration (FDA) criteria for drug approval require substantial evidence of clinical benefit from adequate and well-controlled trials (1). Efficacy should be demonstrated by either prolonging patient survival or improving quality of life (QoL), or both. There is ongoing cont…
Dec 13th • 7 mins read
Clinical Trials in Oncology and Defining Benefit
In the editorial “The Evolution of Clinical Trials in Oncology: Defining Who Benefits from New Drugs Using Innovative Study Designs”, Goldberg nicely discuss many changing aspects of clinical trial design in oncology. However, I believe their title is misleading. No study will defin…
Jan 22nd • 1 min read

AI-powered real-world evidence: Strategically enhancing value and access
Real-world evidence (RWE) is a critical tool for understanding how medicines perform in diverse patient populations and real-world settings. However, the sheer volume and complexity of RWE data pose challenges for traditional analysis methods. Artificial intelligence (AI) is revolutionizing the pha…
Aug 22nd • 5 mins read
