Results for 'T cell'

Job Search Checklist for Aspiring Medical Science Liaisons
Medical Science Liaison, MSL role, MSL career tips, Pharmaceutical industry jobs, Break into MSL career
Tips to Help Break into Your First MSL Role The Medical Science Liaison (MSL) role, has become one of the most sought-after career paths in the Pharmaceutical and Biotech industry. What does it take to land a role as a Medical Science Liaison? As an MSL Recruiter, this is a question that I am…
May 17th • 2 mins read

How to Prepare for a Virtual or Video Interview
virtual interview tips, video interview preparation, mastering virtual communication, professional video interview setup, successful online interview
As we are quickly becoming thrust into the age of “virtual” meetings and video interviews, job seekers and industry professionals need to be completely prepared to master this new paradigm of interpersonal communication. Here are some important tips for acing your next video interview: …
Mar 19th • 1 min read

Presentation Nails and Fails: 7 Tips to Ace Your Next MSL Presentation
MSL interview tips, scientific presentation skills, hiring managers expectations, medical science liaison communication, virtual interview preparation
The presentation portion of the MSL interview is truly “make or break,” and more often than not, the most highly weighted category of the entire interview process. Countless Hiring Managers have passed on MSL candidates that do not perform well on their scientific presentation – re…
Oct 12th • 1 min read

Combatting the “Summer Slow Down" – MSL Job Search Tips for Slower Months
Medical Science Liaison, Medical Affairs recruitment, job search strategies, networking in medical field, summer hiring trends
There is definitely a cyclical nature to hiring Medical Science Liaisons and Medical Affairs professionals. Typically, peak season for recruitment and hiring is from March to June, which is the time-frame in and around which MSLs collect their annual bonuses. However, Medical Science Liaison hiring …
Jun 23rd • 2 mins read

Clinical Educator Oncology Program Improves Adherence by 29%
CASE STUDY (US) The Challenge Client’s specialty product had a complicated oral therapy regimen requiring supplemental support to patients. Therapy administered for chronic life-threatening condition in which patients are generally older and polypharmic. Our Soluti…
Sep 3rd • 1 min read

Transforming oncology: Five frontiers driving progress in cancer care
From biomarker-driven breakthroughs to AI-powered early detection and a renewed commitment to equity and patient centricity, the past 12 months have seen major strides across cancer research, treatment, and communication. At Inizio, we’ve had a front-row seat to this transformation, supporting…
May 16th • 5 mins read

Are Quality of Randomized Clinical Trials and ESMO-Magnitude of Clinical Benefit Scale Two Sides of the Same Coin, to Grade Recommendations for Drug Approval?
quality randomized studies, ESMO-MCBS, drug approval
The approval of new cancer drugs by the FDA and EMA is primarily based on positive results from well-designed randomized phase III clinical trials (RCTs). Not all RCTs are analyzed to support drug approval recommendations, highlighting the need for scales to evaluate RCT quality and clinical…
Feb 11th • 3 mins read

Does biomarker use in oncology improve clinical trial failure risk? A large-scale analysis
biomarker, clinical trial, failure, analysis, NSCLC, oncology
Data from clinical trials across four oncology indications (breast cancer, NSCLC, melanoma, and colorectal cancer) from 1998 to 2017 were analyzed to assess drug approval likelihood based on biomarker status. Using multi-state Markov models, which describe stochastic processes, the study…
Feb 23rd • 8 mins read
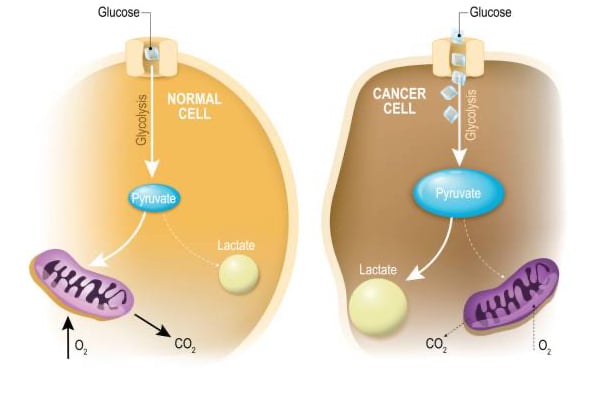
“Oncometabolism: The switchboard of cancer: An editorial”
Otto Warburg, biochemist, FDA, FDG-PET, MRS, Oncometabolism
Otto Warburg's discovery in the 1920s highlighted a unique bioenergetic phenotype in tumor cells, emphasizing their reliance on lactic acid fermentation, which spurred the field of oncometabolism. Oncometabolism has led to advancements like FDG-PET imaging for cancer diagnosis and staging,…
Feb 1st • 1 min read
Loose Regulatory Standards Portend a New Era of Imprecision Oncology
precision oncology, TMB-high, homologous recombination deficiency, Olaparib, pembrolizumab
Precision oncology aims to tailor cancer treatment based on genetic understanding, revolutionizing oncology. The FDA has been approving drugs under precision oncology with broad indications that may not align with studied populations. Examples include the approval of pembrolizumab for a wide ran…
Dec 1st • 4 mins read

Safeguarding cancer research funding by European charities amidst the COVID-19 pandemic
cancer charities, cancer research, COVID‐19, funding, pandemic, policy, AECC, AIRC
The COVID-19 outbreak has had a significant impact on cancer research and cancer care. European cancer charities are required to reassess their strategies. There is a need to safeguard income and provide support to cancer researchers. It is crucial to sustain cancer research funding during thes…
Nov 22nd • 3 mins read
Quantitative Clinical Pharmacology of T‐Cell Engaging Bispecifics: Current Perspectives and Opportunities
quantitative clinical pharmacology, T-Cell, TDB, bispecificss
The pharmacological response of TDBs involves complex interactions between T-cells, tumor cells, and TDBs, posing challenges in understanding pharmacokinetics, tissue distribution, target engagement, and exposure-response relationships. Dosing strategy is crucial for determining the therapeu…
Nov 18th • 15 mins read
Access to Novel Drugs for Non-Small Cell Lung Cancer in Central and Southeastern Europe: A Central European Cooperative Oncology Group Analysis
Health Outcomes and Economics of Cancer Care, Lung Cancer, NSCLC, EMA
Treatment for non-small cell lung cancer (NSCLC) has significantly improved with the introduction of targeted and immune-oncologic drugs. Despite rapid development and European Medicinal Agency (EMA) registration, these novel drugs are not easily accessible in Central and Eastern European (CEE) c…
Nov 24th • 10 mins read
Rise of Antibody-Drug Conjugates: The Present and Future
Antibody-drug conjugates (ADCs) are designed for targeted cancer therapy by delivering cytotoxic agents specifically to tumor cells, minimizing harm to normal cells. ADCs have evolved significantly since the first FDA approval in 2000, with advancements leading to approvals for various tumor type…
May 25th • 20 mins read

The pitfalls and promise of liquid biopsies for diagnosing and treating solid tumors in children: a review
Liquid biopsies, Pediatric solid tumors, Cell-free DNA profiling
Cell-free DNA profiling via blood samples is emerging as a non-invasive method for cancer genomic characterization. Liquid biopsies are set to be integrated into pediatric cancer clinical trials, offering benefits such as earlier therapy response monitoring and detection of residual disease.…
Jan 3rd • 10 mins read
Publication statuses of clinical trials supporting FDA-approved immune checkpoint inhibitors: a meta-epidemiological investigation
Anticancer drugs, Clinical trials, Drug approval, Immune checkpoint inhibitors, Publications, United states food and drug administration
The study investigates the publication status of clinical trials for anticancer drugs approved by the FDA, focusing on immune checkpoint inhibitors (ICPis). Data from ICPis approved between 2011 and 2014 was analyzed, revealing that 58% of ICPis trials were published within two years …
Oct 24th • 18 mins read

The rise of oncology biosimilars: from process to promise
biologic, biosimilar, breast cancer, SB3, trastuzumab
Biosimilars are biologic products that are highly similar to the approved originator molecule, with no clinically meaningful differences. They are expected to play a key role in cancer treatment by reducing costs and improving access. Regulatory bodies use robust mechanisms for approval, involvi…
Aug 23rd • 18 mins read
Early-drug development in the era of immuno-oncology: are we ready to face the challenges?
immunotherapy, early-drug development, toxicology studies, dose determination
The classical development of drugs is being replaced by a seamless drug-development process. First-in-human trials now often include large expansion cohorts to quickly identify early signs of activity and achieve rapid regulatory approval. Intense competition among pharmaceutical companies and t…
Jun 26th • 26 mins read

Biosimilars in Oncology: Latest Trends and Regulatory Status
oncology, biologics, biosimilars, regulatory framework, traceability
Biologic-based medicines are crucial in treating various diseases, particularly cancer, and hold a significant portion of the global pharmaceutical market. Biosimilars, which are highly similar to biologic drugs but not identical, offer potential benefits such as enhanced access and cost savings,…
Dec 5th • 20 mins read

Medical affairs: The power behind redefining commercialization
Medical affairs commercialization, integrating science in pharma, redefining medical affairs roles, strategic medical affairs leadership, early engagement with payers, pharmaceutical commercialization strategies, upskilling in medical affairs, medical aff
In March 2024, Envision Pharma Group held a Medical Affairs Professional Society (MAPS) Americas roundtable with leaders discussing the role of science in commercialization strategies and the importance of integrating robust scientific understanding into the commercial journey. Envision revisite…
Aug 2nd • 3 mins read
