Results for 'FDA'
Browse oncology articles matching FDA — expert analysis, clinical perspectives, and industry updates from across drug development and care delivery. Use the tabs above for podcasts, press releases, partners, and people for the same search.

Evaluation of Trials Comparing Single-Enantiomer Drugs to Their Racemic Precursors: A Systematic Review
single-enantiomer racemic drug pairs, single-enantiomer, FDA, RCTs, generic drugs
Chiral Switching: A strategy where drug manufacturers develop a single-enantiomer formulation from a racemic one to extend market exclusivity, often without proving enhanced efficacy or safety. Objective: To evaluate randomized clinical trials (RCTs) comparing FDA-approved single-enantiomer drugs…
May 6th • 18 mins read
Comparative study on anticancer drug access times between FDA, EMA and the French temporary authorisation for use program over 13 years
Innovation, Expanded access, Early drug access, Cancer, FDA, EMA
Cancer incidence is increasing globally, and while medical innovation significantly impacts patient survival, the drug development process is lengthy, often exceeding 10 years for marketing authorization (MA). France has implemented the ATU (Temporary Authorization for Use) program to facil…
Apr 7th • 12 mins read

Assessment of Coverage in England of Cancer Drugs Qualifying for US Food and Drug Administration Accelerated Approval
FDA, NHS, accelerated approval, NICE, clinical trial findings
The study evaluates the acceptance and coverage of FDA-accelerated approved cancer drugs by the National Health Service (NHS) in England, focusing on decisions made by the National Institute for Health and Care Excellence (NICE). A total of 93 cancer drug indications received accelerated approval…
Feb 22nd • 10 mins read

Assessment of Food and Drug Administration- and European Medicines Agency-Approved Systemic Oncology Therapies and Clinically Meaningful Improvements in Quality of Life: A Systematic Review
ESMO, MCBS, FDA, ASCO-VF, EMA, QOL
Recent oncology therapies approved by the FDA and EMA often lack evidence of clinically meaningful improvements in quality of life (QOL). Only 40% of FDA-approved and 58% of EMA-approved oncology therapies had published QOL evidence. Clinically meaningful QOL improvements beyond minimal dif…
Feb 11th • 4 mins read
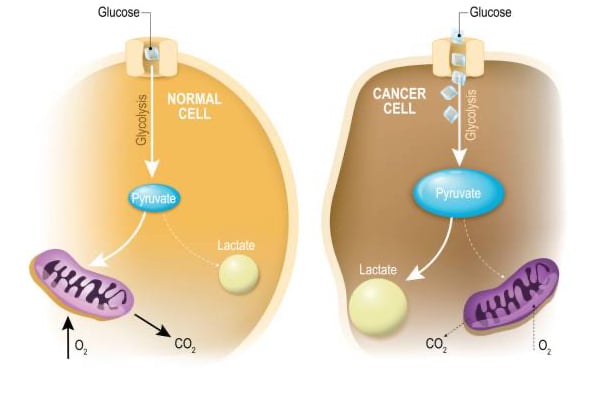
“Oncometabolism: The switchboard of cancer: An editorial”
Otto Warburg, biochemist, FDA, FDG-PET, MRS, Oncometabolism
Otto Warburg's discovery in the 1920s highlighted a unique bioenergetic phenotype in tumor cells, emphasizing their reliance on lactic acid fermentation, which spurred the field of oncometabolism. Oncometabolism has led to advancements like FDG-PET imaging for cancer diagnosis and staging,…
Feb 1st • 1 min read

Clinical benefit and cost of breakthrough cancer drugs approved by the US Food and Drug Administration
USFDA, ESMO-MCBS, NCCN, ASCO-CRC, clinical, drug aroval
The study evaluates the clinical benefit and pricing of breakthrough-designated versus non-breakthrough-designated cancer drugs. The analysis covers approvals from July 2012 to December 2017, using frameworks like ASCO-VF, ASCO-CRC, ESMO-MCBS, and NCCN Evidence Blocks. High clinical benef…
Jul 22nd • 12 mins read

The regulatory landscape of precision oncology laboratory medicine in the United States - Perspective on the past 5 years and considerations for future regulation
Oncology, Regulation, Testing, LDTs, FDA, CLIA, CMS
The regulatory landscape for precision oncology in the United States is complex, involving multiple governmental agencies with varying jurisdictions. Since 2014, several regulatory proposals have been introduced following the FDA's draft guidance on laboratory-developed tests. There are ongoing …
May 22nd • 8 mins read

Assessment of Clinical Trials Supporting US Food and Drug Administration Approval of Novel Therapeutic Agents, 1995-2017
clinical trials, FDA, Novel therapeutic agents, biologics
The number of special regulatory programs for drug and biologic approvals by the FDA has increased since the introduction of the Fast Track designation in 1988. This study analyzed 273 new drugs and biologics approved by the FDA for 339 indications across three periods: 1995-1997, 2005-2007,…
Apr 21st • 20 mins read

Patient burden and clinical advances associated with post approval monotherapy cancer drug trials: a retrospective cohort study
clinical advances, monotherapy cancer drug trials, FDA
Objective: The study investigates the efforts to extend the uses of new drugs by testing them for new, non-approved indications and examines the patient burden and clinical impact. Design and Setting: A retrospective cohort study focused on post-approval trials of anticancer drugs approved betwee…
Feb 17th • 7 mins read
In Vitro-to-In Vivo Extrapolation of Transporter Inhibition Data for Drugs Approved by the US Food and Drug Administration in 2018
In Vitro-to-In Vivo, Transporter Inhibition Data, Drug Approval, FDA, 2018
The analysis focuses on inhibition transporter data from New Drug Applications approved by the FDA in 2018, involving 42 drugs. Key points include: In vitro-to-in vivo predictions were available for nine recommended transporters. 29 parent drugs and 16 metabolites inhibited at least one transpor…
Jan 25th • 12 mins read

Prediction of Drug Approval After Phase I Clinical Trials in Oncology: RESOLVED2
clinical trials, RESOLVED2, FDA
Challenge in Oncology Drug Development: The field is currently facing an increase in the number of antineoplastic agents (ANAs) entering phase I clinical trials (P1CTs) and a high attrition rate for final FDA approval. Objective: Development of a machine learning algorithm, RESOLVED2, to …
Sep 20th • 12 mins read

Cost per Event Averted in Cancer Trials in the Adjuvant Setting From 2018 to 2022
anticancer drugs, adjuvant therapy, FDA approvals, cost per event averted, surrogate endpoints, oncology drug costs
Importance: Evaluating the cost-effectiveness of adjuvant therapies based on the cost per event averted. Objective: To assess the costs per event averted for anticancer drugs approved by the FDA between January 2018 and March 2022. Design, Setting, and Participants: Cross-sectional study of F…
Jun 10th • 30 mins read

Comparison of Long-term Survival Benefits in Trials of Immune Checkpoint Inhibitor vs Non-Immune Checkpoint Inhibitor Anticancer Agents Using ASCO Value Framework and ESMO Magnitude of Clinical Benefit Scale
immunotherapy-triggered LTP, RCTs, FDA, ICI
Importance: Anticancer agents, especially immune checkpoint inhibitors (ICIs), have shown potential for long-term durable survival in some patients. However, traditional clinical benefit measures may not accurately capture this, leading to proposed amendments in valuation frameworks. Objectives: …
Jul 10th • 12 mins read
Audit of Data Sharing by Pharmaceutical Companies for Anticancer Medicines Approved by the US Food and Drug Administration
IPD sharing, clinical trial transparency, FDA anticancer approvals, oncology trials, data accessibility, pharmaceutical industry
The study examines the eligibility for individual participant data (IPD) sharing from clinical trials that supported the FDA approval of anticancer medicines over the past 10 years. Of the 304 trials analyzed, 136 (45%) were eligible for IPD sharing, while 168 (55%) were not. IPD sharing rates v…
Jul 28th • 20 mins read

Overall Survival Benefits of Cancer Drugs Approved in China From 2005 to 2020
cancer drug therapy, overall survival, FDA approvals, surrogate endpoints, EMA approvals, quality of life
The study evaluates the overall survival (OS) benefit of cancer drugs approved in China between 2005 and 2020. Out of 141 cancer drug indications approved, 68 showed a documented overall survival benefit, while 34 did not prolong life. Recent regulatory reforms in China aim to improve the availa…
Aug 10th • 30 mins read

Recent Trends in Medicaid Spending and Use of Drugs With US Food and Drug Administration Accelerated Approval
FDA accelerated approval, Medicaid spending, prescription drug costs, surrogate endpoints, cancer drugs, drug approval pathway
The study examines the impact of drugs approved through the FDA’s accelerated approval program on state Medicaid spending. From 1992 to 2020, 216 drug-indication pairs were granted accelerated approval, with a significant increase in cancer drug approvals in recent years. Although drugs wi…
Oct 8th • 25 mins read

FDA validation of surrogate endpoints in oncology: 2005–2022
FDA drug approval, surrogate endpoints, oncology drugs, overall survival correlation, clinical trials analysis, drug approval guidelines
ere are summary bullets based on the provided text: The FDA has increasingly approved oncologic drugs based on surrogate endpoints, which often lack a demonstrated correlation with clinically meaningful outcomes like overall survival. A review of FDA analyses from 2005 to 2022 examined the valid…
Dec 1st • 20 mins read

Association Between US Drug Price and Measures of Efficacy for Oncology Drugs Approved by the US Food and Drug Administration From 2015 to 2020
Cancer drug pricing, Cancer care costs, Cancer drug efficacy, Progression-free survival, Value-based pricing in oncology, FDA anticancer approvals
The US has worse cancer-related outcomes compared to other high-income countries and has the highest cost of cancer care globally. High costs may be attributed to the improved efficacy of expensive new cancer drugs, though the relationship between cost and benefit is debated. A study found a lin…
Oct 31st • 10 mins read
Exposure to US Cancer Drugs With Lack of Confirmed Benefit After US Food and Drug Administration Accelerated Approval
US, FDA, approval, drugs, benefits, cancer
Among 5 oncology indications, 26.1% of eligible treatment initiations involved an Accelerated Approval (AA) indication that was later withdrawn due to lack of benefit. There is an inherent trade-off between expediting access to promising cancer drugs and the potential withdrawal of some indicatio…
Dec 8th • 2 mins read

Evaluating External Validity of Oncology Biosimilar Safety Studies
biologics, external validity, FDA, biosimilars, trastuzumab
Biologics are the fastest growing medication class in the US, significantly contributing to healthcare costs. The Biologics Price Competition and Innovation Act of 2009 created an abbreviated approval pathway to make biological products more accessible. A phase 3 randomized trial of SB3, a trast…
Apr 6th • 2 mins read
