Results for 'imaging'
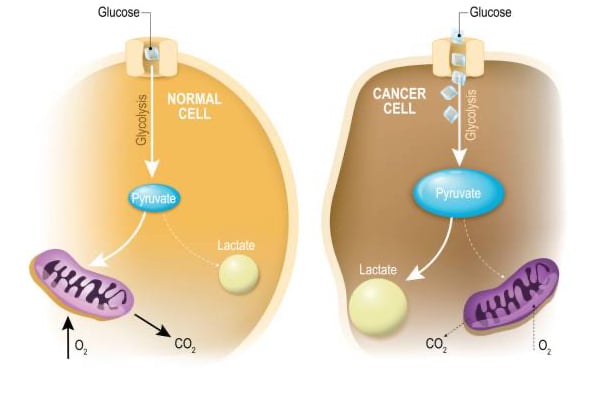
“Oncometabolism: The switchboard of cancer: An editorial”
Otto Warburg, biochemist, FDA, FDG-PET, MRS, Oncometabolism
Otto Warburg's discovery in the 1920s highlighted a unique bioenergetic phenotype in tumor cells, emphasizing their reliance on lactic acid fermentation, which spurred the field of oncometabolism. Oncometabolism has led to advancements like FDG-PET imaging for cancer diagnosis and staging,…
Feb 1st • 1 min read

Preliminary Attainability Assessment of Real-World Data for Answering Major Clinical Research Questions in Breast Cancer Brain Metastasis: Framework Development and Validation Study
real-world data, preliminary attainability assessment, observational study, clinical data warehouse, PAR framework, brain metastasis, breast cancer
The study proposes the PAR framework for data attainability screening in clinical research, particularly useful for addressing unmet clinical needs in BCBM. A survey showed clinical questions had a mean score of 4.37, indicating the significance of these questions. RWE generation is crucial…
Oct 9th • 4 mins read

FDA Acceptance of Surrogate End Points for Cancer Drug Approval: 1992-2019
FDA, Surrogate End Points, Cancer drug approval
194 approvals were analyzed, with 64 (32.9%) relying on surrogate endpoints for the first time in a specific cancer type. Surrogate endpoints often have weak or unknown correlations to overall survival (OS). Many approvals lack strong scientific or regulatory justification, indicating a higher t…
Mar 9th • 4 mins read
Overview of Oncology and Hematology Drug Approvals at US Food and Drug Administration Between 2008 and 2016
drug approval, medical oncology, united states food and drug administration, hematology, surrogate endpoints, marketing, accelerated approval
Background: An overview was conducted of oncology products reviewed by the Office of Oncology Drug Products (OODP) at the FDA for marketing approval between July 2005 and December 2007. The study aims to understand the impact of post-2007 changes in laws, regulations, and organizational structure …
Aug 4th • 15 mins read
